Characterization of Triterpenoid Saponins, Phenylethanoids and Flavonoids in Bacopa Caroliniana (Walt.) B.L. Robins Using LC-ESI-QTOF-MS/MS Technique
Abstract
This research reports upon the chemical constituent of the aerial part of Bacopa caroliniana identified using the liquid chromatography with electrospray ionization/mass spectrometry (LC-ESI-QTOF-MS/MS) technique. The LC separation along with high resolution mass spectrometry data revealed significant differences in phytochemicals of B. caroliniana. The mass spectral fragmentation patterns in both positive and negative modes indicated the numbers, positions, and linkages of sugar moieties in the saponin structures. The distinct m/z 455 [Aglycone + H-H2O]+ from in source fragmentation suggested that the triterpenoid saponins in this plant are jujubogenin type whereas m/z 473 [Aglycone + H]+ are pseudojujubogenin type. This fragmentation pattern led to identification of bacoside A3 (1), bacopaside X (2) and their derivatives (3-6). The four derivative compounds (3-6) are differed in the glycosidic part with the malonyl moiety conjugated to the hexose or/and pentose moieties. Additionally, four phenylethanoid glycosides e.g.acteoside (I), acetylacteoside (III) and two of their isomeric forms (II, IV) were identified. Nine flavonoids (a-i), two flavone and seven O-methoxy flavonoids were also reported. All the identified compounds were reported for the first time in B. caroliniana. This chemical diversity can be very important as supporting data for further uses in pharmaceutical and food industries.
Keywords
Bacopa caroliniana, LC-ESI-QTOF-MS/MS, Triterpenoid saponins, Phenylethanoid glycosides, Flavonoids, Structure elucidation
Introduction
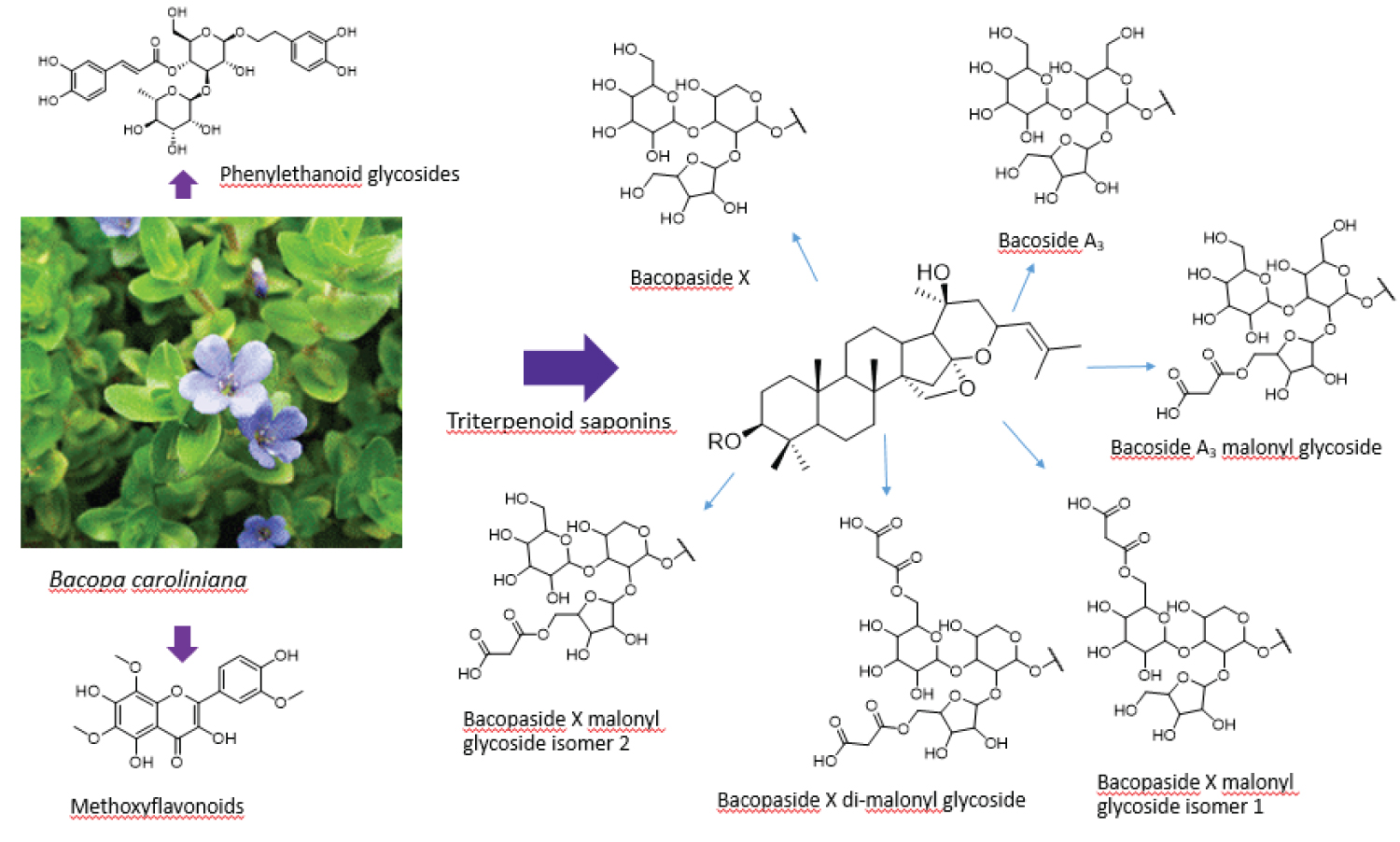
Bacopa caroliniana (Walt.) B.L. Robins or Lemon bacopa (Plantaginaceae) is a perennial aquarium plant. The leaves of this plant are succulent and release smell like the lemon-mint when disturbed and the flowers are bright blue [1]. In Thailand, there are three species of Bacopa genus, i.e. B. monnieri, B. caroliniana and B. floribunda [2]. B. monnieri (Brahmi) has traditionally been reported as a herbal medicine in Ayurvedic medicine for memory improvement [3]. The compounds responsible for memory enhancing effects of Brahmi are triterpenoid saponins i.e. bacoside A3, bacopaside I, bacopaside II, bacopasaponin C and bacopaside X [4,5]. The investigation of triterpenoid saponins has drastically increased due to their diverse and potentially attractive biological activities. B. caroliniana, one of bacopa species, might be a source of saponin glycosides in the same fashion as B. monnieri. In addition, B. caroliniana possessed significant activity against microorganisms, especially Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans [6]. The eighteen volatile compounds from B. caroliniana have been identified and tested on the inhibition of acetylcholinesterase and insecticidal activities. It was found that α-terpinolene exhibited the highest activities [7]. In the recent report, methanolic extractfrom aerial part of B. caroliniana showed moderated anti-lipid peroxidation activity [8].
Previously, we have reported a great number of saponins found in B. monnieri [8,9]. In this study, we aim to search for new saponins and other compounds in B. carolianiana using LC-ESI-QTOF-MS/MS method. This method is a powerful tool for the analysis of complex mixtures, especially those for crude extracts of natural products. A method is using simultaneous acquisition of exact masses at high and low collision energies, to obtain fragmentation information allowing the elucidation of elemental composition and structural identification of compounds. The results of this study for finding new saponin and other chemical constituents could provide new opportunities to promote this plant which possesses high amounts of bioactive compounds in the pharmaceutical and food industries.
Experimental Section
Chemicals and plant materials
Acetonitrile (ACN) and methanol (MeOH) with LC-MS grade were purchased from Lab scan (RCI Labscan, Thailand). Formic acid was bought from Merck (Darmstadt, Germany). Water type I was purified by a Milli-Q purification system from Millipore (Bedford, MA, USA).
The authentic compounds: Bacoside A3 and bacopaside X were purchased from Natural Remedies Pvt. Ltd. (Bangalore, India). Luteolin and apigenin were obtained from Chromadex (CA, USA).
B. caroliniana was planted in January - July 2017 at Faculty of Pharmaceutical Sciences, Naresuan University. This plant was identified by Assist. Prof. Dr. Pranee Nangngam and its voucher specimen (Saesong013) have been deposited at Department of Biology, Faculty of Science, Naresuan University. The aerial part (10 cm from the top) of the sample was collected based on a previous method [10]. The plant was cleaned, and oven dried at 50 ℃ for 24 h. After that, the plants were ground and passed through a 60 mesh sieve and stored in airtight plastic containers under refrigeration at -20 ℃ for later use.
Sample preparation
A 20 mg pulverized sample was added to a plastic tube and was then extracted with 2.0 mL of 70% (v/v) methanol/water for 20 sec on a Vortex Genie 2 mixer (Scientific Industries, New York, USA) and followed by 15 min extraction in an ultrasonic bath (Grant MXB14, Keison Products, UK). Prior to LC-MS analysis, the extract was filtered through a Nylon 0.22 µm, 13 mm syringe filter (ANPEL Laboratory Technologies, Shanghai Inc.).
LC-ESI-QTOF-MS/MS analysis
Chromatographic experiments were performed with an Agilent 6540 Q-TOF-MS spectrometer (Agilent Technologies, Singapore) coupled with an Agilent 1260 Infinity Series High performance liquid chromatography (HPLC) system (Agilent, Waldbronn, Germany). The separation was performed with the ZORBAX Eclipse plus C18, 4.6 mm × 100 mm, 3.5 µm column (Agilent Technologies, USA) at a flow rate of 600 µL/min and the column temperature was set to 35 ℃. The mobile phase A was water type I and B was acetonitrile. Both phases contained 0.1 % (v/v) formic acid. The gradient mode started with 35% solvent B for 0-8 min, 35-38% B for 8-10 min, 38-50% B for 10-14 min, 50-80% B for 14-25 min and post-run for 5 min. The injection volume was 20 µL. The operating parameters for MS detection were as follows: drying gas (N2) flow rate 10.0 L/min; temperature 350 ℃; nebulizer pressure 30 psig; capillary 3500 V; skimmer 65 V; octapole RFV 750 V; and fragmentor voltage 250 V in negative mode and 100 V in positive mode. The mass range was set at m/z 100-1200 with a 250 ms/spectrum. The non-target MS/MS mode was set up at three collision energies of 10, 20, and 40 V using high purity nitrogen gas. All acquisition and analysis of the data were controlled by MassHunter Data Acquisition Software version B.05.01 and MassHunter Qualitative Analysis Software B 06.0 respectively (Agilent Technologies, USA). Analysis of each sample was performed both in positive and negative ionization modes including non-targeted MS/MS mode to provide abundant information for structural identification.
Results and Discussion
Structure elucidation of triterpenoid saponins
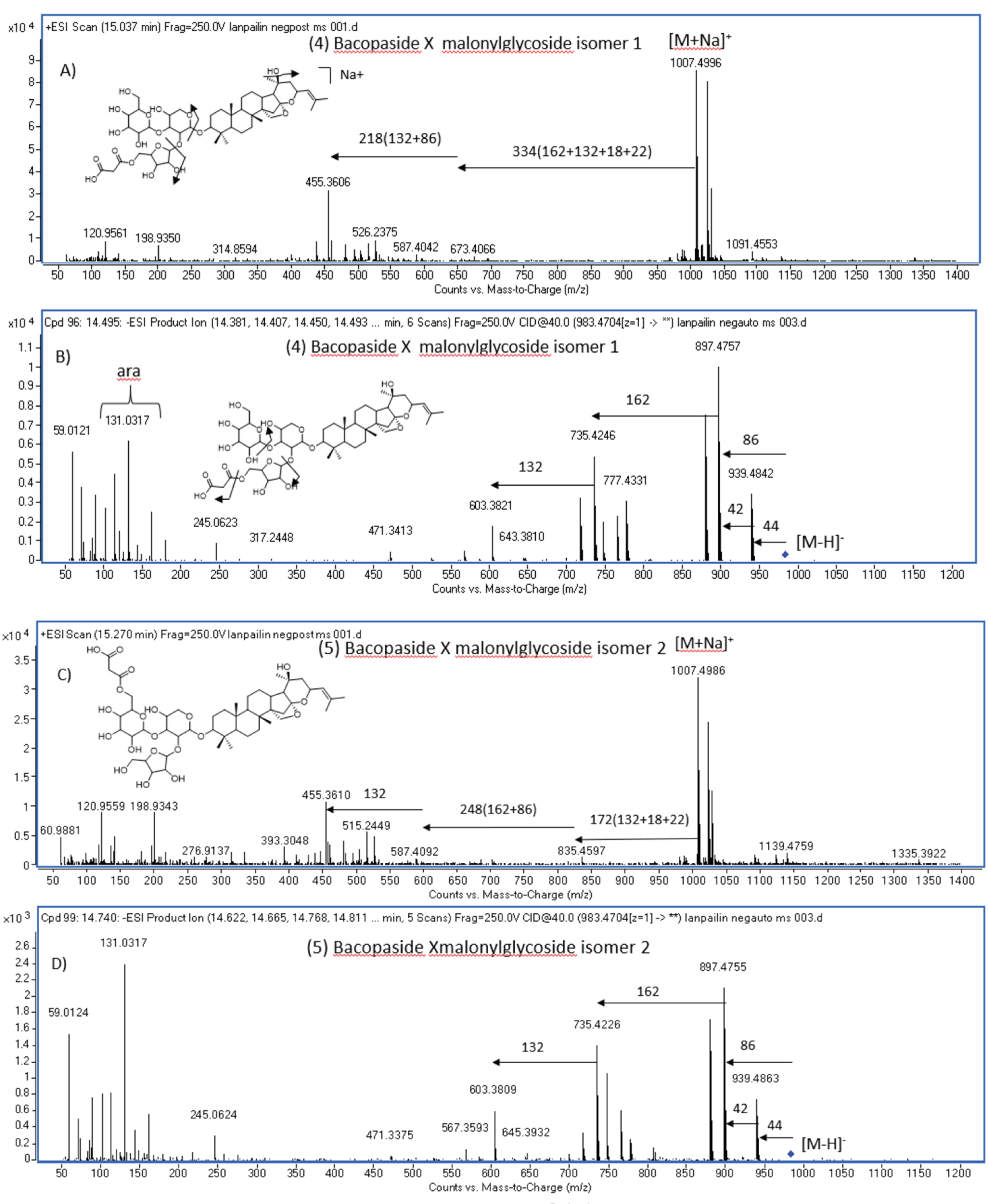
HPLC coupled to ESI-QTOF-MS/MS (tandem mass spectrometry) was applied to separate, characterize and identify triterpenoid saponins (1-8) in B. caroliniana methanolic extract. The previous work indicated that jujubogenin glycosides and pseudojujubogenin glycosides commonly found in Bacopa species can be discriminated by the characteristic of in source fragmentation at m/z 455 [aglycone + H-H2O]+ of jujubogenin glycosides and m/z 473 [aglycone + H]+ of pseudojujubogenin glycosides [9]. The ESI-QTOF-MS/MS data of B. caroliniana shown at m/z 455 [aglycone + H-H2O]+ in compounds 1- 6 proposed the presence of jujubogenin type in this plant.
Furthermore, in negative ion mode of MS/MS fragmentation, saponins under went glycosidic cleavages using high energy (CE 40 V) resulting in sequential loss of sugar moieties leaving one sugar unit attached to aglycone with the lowest m/z peak. The sugar attached at C3 position would be glucose if the typical of m/z 633; 471 + 162; [aglycone + glucose]- was observed or arabinose if m/z 603; 471+132; [aglycone + arabinose]- was present. The neutral loss of sugar; glucose (m/z162) and arabinose/xylose (m/z 132) can be seen in the hydration form, loss of 18 Da (-H2O) due to the conjugation process. The aglycone-sugar linkage of glucose could be confirmed by m/z 161,113,101, while that of arabinose could be observed at m/z 131, which were highly abundant in the left side of the mass spectrum (Figure 1B and Figure 1D). So clearly define that the sugar moiety link to aglycone is arabinose. Consequently, identification of saponins in Bacopa species based on both positive and negative characteristic fragmentation patterns were successfully conducted (Table 1).
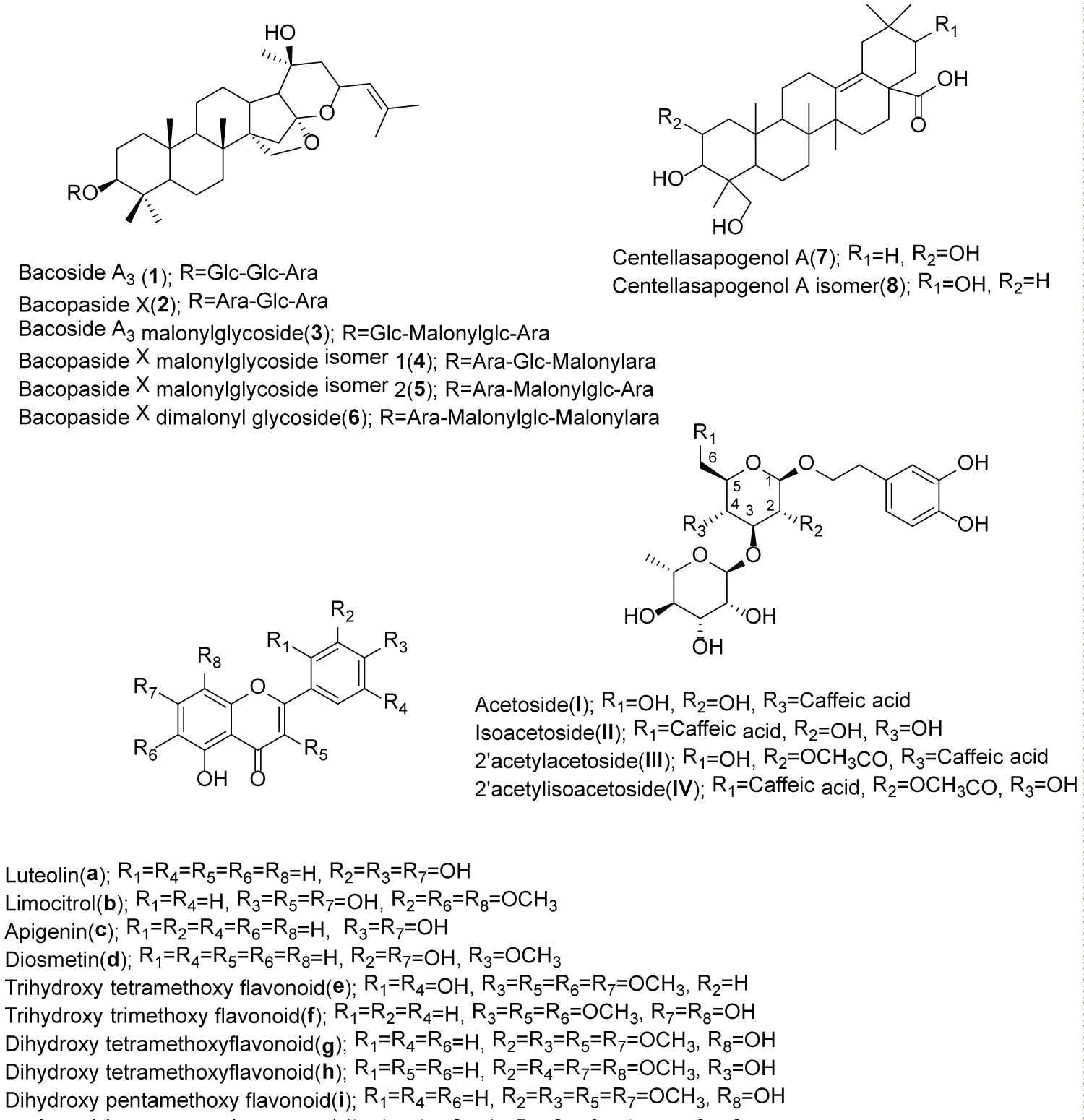
Compounds 1 and 2 were elucidated using the above-mentioned characteristic fragmentations and confirmed with the reference standards as bacoside A3 and bacopaside X, respectively. These two compounds have been found in B. monnieri [9].
Compound 3 showed m/z 1037.5103[M + Na]+, which gained 86 Da higher than that of bacoside A3. Its fragmentation in positive mode showed loss of 248Da (162 + 86: Glucosyl + malonyl). It is likely that this malonyl group is attached at the 6-position of the glucosyl moiety. In negative mode, the loss of 44Da (CO2), 42Da (CH2=CO) and 60Da (CH3COOH) represented the typical loss of malonyl saponin [11]. This compound was, therefore, proposed to be bacoside A3 malonylglycoside.
Compounds 4 and 5 with the same m/z 983.4861 [M-H]- and 1007.4996 [M + Na]+ in negative mode showed the loss of 44 and 42 Da implying the presence of a malonyl moiety in their structures. In positive mode, the sequential loss of 86 Da in compound 4 occurred together with the loss of xylosyl (218Da = 132 + 86), where as compound 5 malonyl group was lost together with glucosyl moiety (248Da = 162 + 86). These two compounds are tentatively identified as bacopaside X malonyl glycoside isomer 1 (jujubogenin 3-O-(5-O-malonyl)-α-L-arabinofuranosyl (1-2)-[β-D-glucopyranosyl(1-3)]-α-L-arabinopyranoside) (4) and bacopaside X malonyl glycoside isomer 2 (jujubogenin 3-O-α-L-arabinofuranosyl (1-2)-[β-D-(6-O-malonyl)-glucopyranosyl (1-3)]-α-L-arabinopyranoside) (5). The difference of mass fragmentation can be seen in (Figure 1). Compound 4 has been previously isolated from ethanol extraction of the stems of Colubrina retusa [12].
Compound 6 with m/z 1069.4865 [M-H]-and 1093.5012 [M + Na]+ showed 86 Da higher than compounds 4,5 and the neutral loss of 88Da (2-CO2) and 42Da (CH2 = CO). The 42Da might be 2 groups of malonyl that conjugated with glucosyl and xylosyl moieties. Thus, this compound was proposed as bacopaside X di-malonylglycoside.
The other two triterpenoid saponins eluted at the retention time 19.4 and 20.5 min (7 and 8) with the same m/z 487.34 [M-H]- and 511.34 [M + Na]+ are tentatively identified as centellasapogenol A and its isomer by comparison with the literature [13]. Without the standard or NMR data, it is still inconclusive whether 7 or 8 is centellasapogenol A.
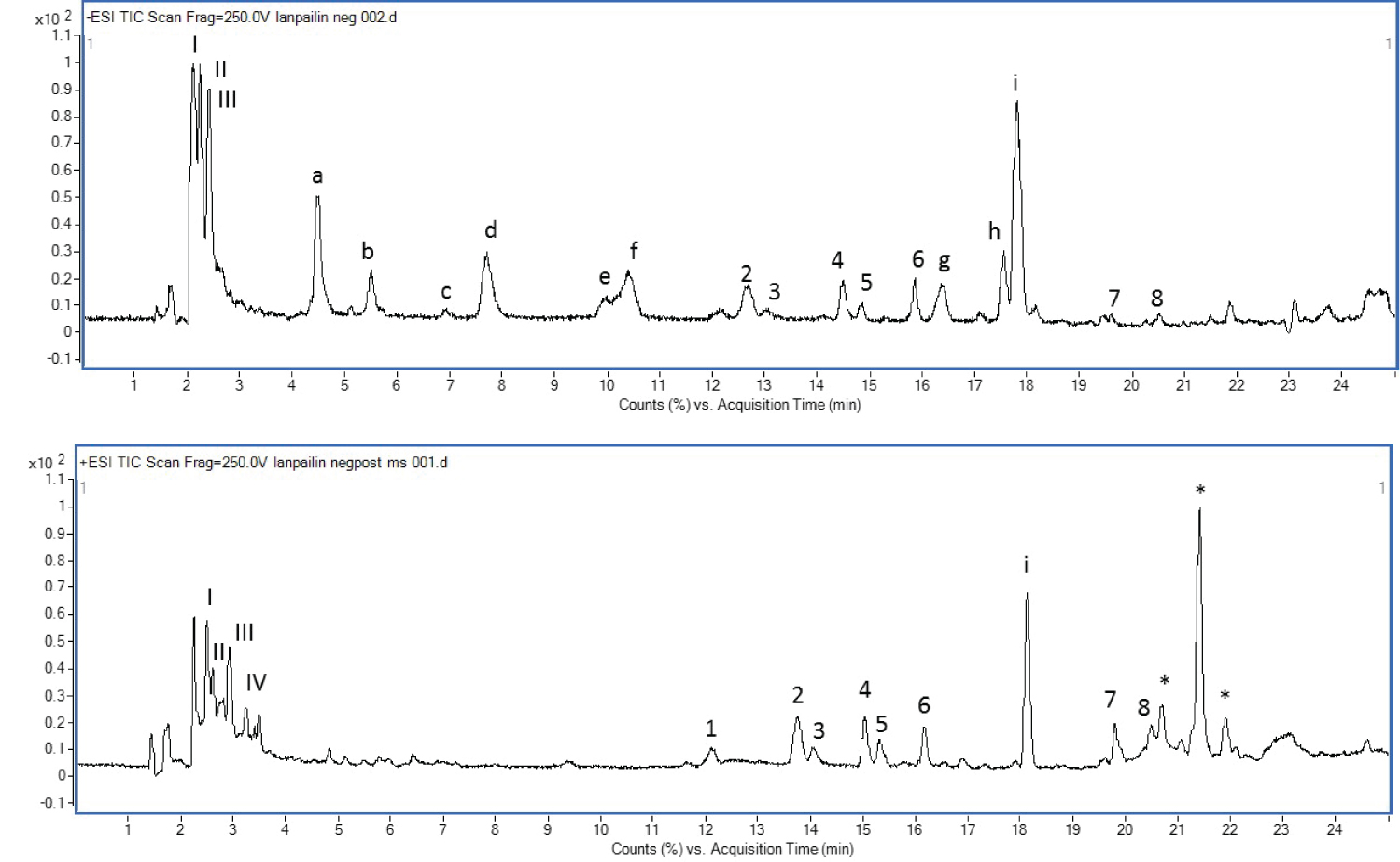
The MS data of triterpenoid saponins are illustrated in Table 1. The total ion chromatogram (TIC) of B. caroliniana is presented in Figure 2.
Structure elucidation of phenylethanoid glycosides
Compounds I and II eluted at the retention time of 1.69 and 2.11 min with the m/z 623 [M-H]-, and 647 [M + Na]+ showed the similar fragmentation patterns, which indicated that they might be isomers (Table 2). In the same fashion, compounds III and IV at 2.25 and 2.42 min were isomers with the m/z of 665 [M-H]- and 689 [M + Na]+. By comparison of MS data with the previous reports, compounds I-IV can be proposed as phenylethanoid glycoside, in which I was acteoside and II-IV were its derivatives. Acteoside (I) (m/z 623.1971) produced the fragment at m/z 461 by the loss of the caffeic acid (162Da) moiety, and then generated an ion at m/z 315 by a further loss of rhamnose (146Da). The dominant fragment at m/z 161 originated from caffeic acid with the loss of water molecule. Isoacteoside (II) showed the same m/z and fragmentation pattern as acteoside but eluted with a longer retention time. The other two compounds (III and IV) with the m/z 665.2100 [M-H]-, 689.2176 [M + Na]+ which were 42Da greater than that of acteoside, were tentatively identified as acetylacteoside and acetylisoacteoside [14]. These compounds were previously found in Cistanches Herba, a famous traditional Chinese medicine from the dried succulent stems of Cistanche deserticola Y. C. Ma and Cistanche tubulosa (Schrenk) Wight. Both herb have been used for the treatment of kidney deficiency, impotence, female infertility, morbid leucorrhea, profuse metrorrhagia, and senile constipation [14,15]. Phenylethanoid glycosides found in B. coroliniana contain one more rhamnose moiety than B. monnieri [8].
Structure elucidation of flavonoids
Besides saponins and phenylethanoid glycosides, flavonoids were also tentatively identified (Table 3). Compounds a and c (Figure 2) with the m/z 285 [M-H]- and 269 [M-H]- were unambiguously assigned as luteolin and apigenin respectively and confirmed with authentic compounds. These two compounds were also found in B. monnieri. Compound d with the m/z 299 [M-H]- was assigned as diosmetin due to containing of 30Da (-OCH3) higher than apigenin. Compound b with the m/z 375 [M-H]- was proposed as a tetrahydroxy trimethoxy flavonoid, which was tentatively identified as limocitrol. Compounds e-I also provided similar fragmentation patterns of flavonoids with the 15Da distinct loss. Therefore, they were proposed as flavonoid derivatives that contain hydroxyl and/or methoxy groups attached to the C atom of the flavonoid backbone (flavone and flavonol). Compound e with m/z 389 [M-H]- was identified as trihydroxy tetramethoxy flavonoid. Compound f with m/z 359 [M-H]- ; 30Da less than compound e may be trihydroxy trimethoxy flavonoid. Compounds g and h showed the same m/z 373 [M-H]-, were proposed as dihydroxy tetramethoxy flavonoid. Compound i with m/z of 403 [M-H]- was identified as dihydroxy pentamethoxy flavonoid. However, the positions of hydroxyl and methoxy group on these compounds cannot be pointed out by this LC-MS/MS information. The compound identities still need proof by other techniques such as NMR. Three compounds eluted at 20.69, 21.40 and 21.9 min are unidentified in this time. Luteolin, apigenin, diosmetin and limocitrol are commonly found in citrus fruit [16]. The bioactive properties of flavonoids have been primarily associated with decreased oxidative stress and free radical damage activity [17] (Figure 3).
Conclusions
High-resolution LC-ESI-QTOF-MS/MS has led to the tentative identification of 21 compounds in B. caroliniana. Eight compounds found belong to triterpenoid saponin group; six of them were bacopa saponin i.e., bacoside A3 and bacopaside X with their derivative that differ in glycosidic part with the malonyl one or two groups conjugated to the hexose or/and pentose moieties. The other two saponin compounds were proposed as centellasapogenol A and its isomer. Beside this, four phenylethanoid glycosides; acteoside and isoacteoside with their acetyl group were elucidated. Additionally, two flavones; luteolin and apigenin were found. Seven O-methoxy flavonoids were also reported in this plant. All the identified compounds were reported for the first time in B. caroliniana.
Declaration of Competing Interest
The authors declare no conflict of interest.
Acknowledgements
This work was supported by the Royal Golden Jubilee PhD Program [PHD/0039/2557 to Tongchai Saesong and Kornkanok Ingkaninan], the Thailand Research Fund [DBG6080005, IRN61W0005], and the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Ministry of Higher Education, Science, Research and Innovation.
References
- Gettys LA, Della Torre CJ (2015) Lemon bacopa: Bacopa caroliniana.
- Tem S (2014) Thai plant names. (edn) The forest herbarium, Royal forest department, Bangkok.
- Mukherjee GD, Dey CD (1966) Clinical trial on Brahmi I. J Exp Med Sci 10: 5-11.
- Singh H, Dhawan BN (1997) Neuropsychopharmacological effects of the ayurvedic nootropic bacopa monnieri linn, Indian J Pharmacol 29: 359-365.
- Russo A, Borrelli F (2005) Bacopa monniera, a reputed nootropic plant: An overview, Phytomedicine 12: 305-317.
- Dulger, Hacioglu N (2009) Antimicrobial activity of bacopa caroliniana, Asian J Chem 21: 4077-4080.
- Liu TT, Chao LKP, Hong KS, et al. (2019) Composition and insecticidal activity of essential oil of Bacopa caroliniana and interactive effects of individual compounds on the activity. Insects 11: 23.
- Saesong T, Allard PM, Queiroz EF, et al. (2019) Discovery of lipid peroxidation inhibitors from bacopa species prioritized through multivariate data analysis and multi-Informative molecular networking. Molecules 24: 2989.
- Nuengchamnong N, Sookying S, Ingkaninan K (2016) LC-ESI-QTOF-MS based screening and identification of isomeric jujubogenin and pseudojujubogenin aglycones in Bacopa monnieri extract. J Pharm Biomed Anal 129: 121-134.
- Phrompittayarat W, Jetiyanon K, Wittaya Areekul S, et al. (2011) Influence of seasons, different plant parts, and plant growth stages on saponin quantity and distribution in Bacopa monnieri, Songklanakarin J Sci Technol 33: 193-199.
- Qi LW, Wang HY, Zhang H, et al. (2012) Diagnostic ion filtering to characterize ginseng saponins by rapid liquid chromatography with time-of-flight mass spectrometry. J Chromatogr A 1230: 93-99.
- Li XC, ElSohly HN, Nimrod AC, et al. (1999) Antifungal jujubogenin saponins from colubrina retusa. J Nat Prod 62: 674-677.
- Azerad R (2016) Chemical structures, production and enzymatic transformations of sapogenins and saponins from Centella asiatica (L.) Urban. Fitoterapia 114: 168-187.
- Li Y, Zhou G, Peng Y, et al. (2016) Screening and identification of three typical phenylethanoid glycosides metabolites from Cistanches Herba by human intestinal bacteria using UPLC/Q-TOF-MS, J Pharm Biomed Anal 118: 167-176.
- Zhang Y, Wang Y, Yang S, et al. (2019) The difference of chemical components and biological activities of the raw products slices and the wine steam-processed product from Cistanche deserticola. Evid Based Complement. Alternat Med 2019: 2167947.
- Ledesma Escobar CA, Priego Capote F, Luque de Castro (2019) Relevance and analysis of citrus flavonoids. In: Watson RR, Polyphenols in Plants. (2nd edn), 133-150.
- Russo A, Acquaviva R, Campisi A, et al. (2000) Bioflavonoids as antiradicals, antioxidants and DNA cleavage protectors. Cell Biol Toxicol 16: 91-98.
Corresponding Author
Nitra Nuengchamnong, Science Laboratory Centre, Faculty of Science, Naresuan University, Phitsanulok, 65000, Thailand, Tel: +66898291285.
Copyright
© 2021 Nuengchamnong N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.