Atelectasis with Torpid Evolution in Patients with Cystic Fibrosis
Background
Cystic fibrosis (CF) is a chronic and progressive disease. Respiratory complications, usually, afect the quality of life, morbidity and prognosis of the disease [1-4]. Within respiratory complications, atelectasis is which the least medical literature has generated, without consensus on its etiology, prognosis and treatment [5]. In a previous study of our group, the incidence was 2.66%, 2.33% were lobar and 1.33% segmental [6]. Among the causes would be the existence of mucus plugs or alterations of the pulmonary architecture in which, the progressive and irreversible damage of the airway could cause distortion, obstruction and bronchial dilatation [5,7-11].
Currently, there are very few studies with a small number of cases that analyze the impact of atelectasis in the prognosis of CF. Atelectasis could be a variable of bad prognosis as a complication in CF. In this paper, we describe eleven cases, whose evolution was affected after being diagnosed with a pulmonary atelectasis, either by dying, entering the waiting list of lung transplant or, finally arriving at a lung transplant. We would like to show that patients suffering from these complications can have a worse prognosis and a lower survival.
Presentation of the Cases
This study is part of a more complex work in which patients with cystic fibrosis who have suffered at least one episode of atelectasis are being analyzed. We analyzed 46 patients who had suffered an episode of atelectasis in multidisciplinary CF units at a national and international level, collected from their beginning until July 2017. The follow-up of the cases has been carried out from the appearance of the complication until, at least, two years later. From this broader work, we have studied the cases of torpid evolution (Table 1, Table 2a and Table 2b).
Results
In our study, 11 out of of 46 patients (23.9%) who had suffered atelectasis during their follow-up, had a torpid evolution. Of these, six patients were women (54.5%). The mean age of the patients at the moment of lung transplantation and their death was 32-years-old.
Five patients died (45.5%), of which one was during the transplant surgery, another on the lung transplant list and three others without being able to access the waiting list for lung transplantation due to the rapidity of the respiratory worsening. Phe508del was the most frequent. Most of them had pancreatic insufficiency (72.7%). With respect to other pulmonary complications, the most striking finding was that three patients had hemoptysis (27.3%), of which two required embolization (18.2%) (Table 1).
According to microbiology at the time of atelectasis, most of them had chronic bronchial infection (81.8%): Pseudomonas aeruginosa stood out in eight of 11 patients (72.7%), followed by methicillin-sensitive Staphylococcus aureus in six of them (54.5%) (Table 1).
Regarding the location of atelectasis, five patients presented it in the right upper lobe, of which one of them evolved to complete pulmonary atelectasis. One patient presented segmental atelectasis and, finally, one patient suffered two episodes of atelectasis. None of them had radiological resolution of the complication (Table 1).
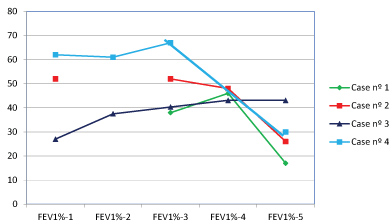
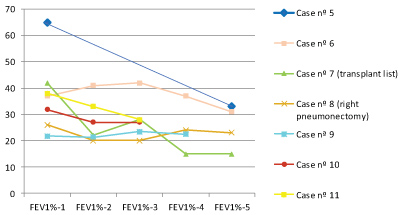
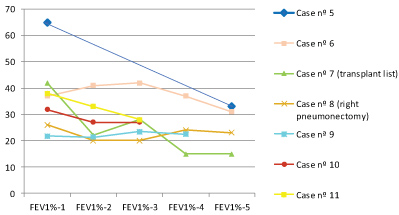
Lung function after the onset of atelectasis had a tendency to fall during the follow-up period (Table 2a, Table 2b, Figure 1 and Figure 2). Although no statistical differences were observed, mean z-scores of forced vital capacity (FVC) and forced expiratory volume in one second (FEV1) decrease three, six and 12 months after the onset of atelectasis. Concerning CF respiratory exacerbations, we also founded a tendecy of increasing the number of exacerbations one year after the episode of atelectasis.
Discussion
Atelectasis are well-recognized pulmonary complications that affect the upper lobes due to mucoid occupation, infectious process or aspiration, being essential to favor the clearance of the airway as quickly as possible to avoid complete lung collapse [2,5,9-11], as we have observed in our work. Some authors considered that there might be a genetic predisposition towards a specific location, especially in children [5,12]. Although the incidence published was 4-11%, the one published by our group was 2.66% [6] .
In the current study, we found that 23.9% presented a poor evolution, which led us to believe that atelectasis could adversely affect the survival of patients with CF, by favoring respiratory infections and a worsening of lung function. The mean age of these patients was lower than those published (more than 40 years) [1]. It is well known that pulmonary exacerbations are associated with a aggravation of the disease [13,14]. In our cases, we have observed an increase in the number of exacerbations, which would explain their torpid evolution.
All the patients in our study, with the exception of one, were carriers of the Phe508del mutation. This mutation is associated with worse pulmonary function, more bronchial infections and other complications, and a worse prognosis [15].
Women presented a greater fall in lung function and showing the dysregulation in the production of mucus influenced by estrogen [13,16]. Despite the fact that, in general, women have a worse prognosis, we have not found a correlation between the atelectasis of torpid evolution and the female gender.
Different risk factors have been postulated in the development of atelectasis increasing the viscosity of the pulmonary mucus, such as chronic bronquial infection, hyperglycemia and hemoptysis [5]. The relationship of ABPA with the etiology of the atectasias is very controversial, since it could favor the development of mucosal impaction [3,6,13,17,18]. Three of our patients had episodes of hemoptysis. The atelectasis and its inflammation could trigger the onset of hemoptysis. But, likewise, the treatment of hemoptysis, when removing physiotherapy and inhaled therapy for its control, could cause them. In our work, all of our patients had a chronic bronchial infection.
The use of the flexible bronchoscope with instillation of substances with mucolytic effect has been described in patients with atelectasis who have not responded to the usual treatment or as a diagnostic method [18], but without There is no available bibliography on its effective value in the treatment of this complication [6,8,19]. Surgery as a treatment is of doubtful value and only justified by the persistence of atelectasis during a prolonged period with worsening of the disease [10]. In our study, a patient needed a right pneumonectomy, which led to clear clinical improvement and good subsequent evolution. Five patients required a fiberoptic bronchoscopy; saline serum was instilled in four of them, adding DNAse in only one case, without achieving radiological improvement.
According to our results of the present study, several risk factors could be established for the appearance of atelectasis with torpid evolution, such as the fact of being a carrier of the Phe508del mutation, taking into account that this mutation correlates with more severe clinical manifestations, location in the upper lobes, having comorbidities associated with CF, as well as presenting previous pulmonary complications, especially chronic bronchial infection and hemoptysis.
Conclusions
We can conclude that atelectasis seems to cause an increase in pulmonary exacerbations and rapid respiratory deterioration, so it is necessary to treat it early, mainly with different measurements to reduce the sputum viscosity. Among the limitations of the work we found: 1) The small number of patients which prevents concluding if the aggravation of these patients is due to the presence of atelectasis or the underlying disease; 2) This study is retrospective; but this would allow us to analyze the data and the characteristics of the patients more quickly. We believe that, after these results, it is necessary to continue analyzing this complication with multicentre studies.
Conflicts of Interest
"The authors declare no conflict of interest". "The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results".
Funding
"This research was funded by [Fundación NeumoMadriD] "REPERCUSIÓN DE LAS ATELECTASIAS COMO COMPLICACIÓN PULMONAR EN LA FIBROSIS QUÍSTICA".
References
- Lerín M, Prados C, Martínez MT, et al. (2014) Cystic fibrosis in adult age. Rev Clin Esp 214: 289-295.
- Máiz L, Baranda F, Coll R, et al. (2001) Diagnóstico y tratamiento de la afección respiratoria en la fibrosis quística. Arch Bronconeumol 37: 316-324.
- Kumar R, Poongadan MN, Singh M (2015) Allergic bronchopulmonary aspergillosis presenting as lobar or total lung collapse. Pneumonol Alergol Pol 83: 144-150.
- Justicia JL, Solé A, Quintana-Gallego E, et al. (2015) Management of pulmonary exacerbations in cystic fibrosis: Still an unmet medical need in clinical practice. Expert Rev Respir Med 9: 183-194.
- Flight W, Hildage J, Web AK (2012) Progressive unilateral lung collapse in cystic fibrosis: A therapeutic challenge. J R Soc Med 105: 44-49.
- Martínez Redondo M, Prados Sánchez C, Salcedo Posadas A, et al. (2017) Características de las atelectasias como complicación pulmonar en la fibrosis quística. Rev Patol Respir 20: 79-87.
- Loeve M, Hop WC, de Bruijne M, et al. (2012) Chest computed tomography scores are predictive of survival in patients with cystic fibrosis awaiting lung transplantation. Am J Respir Crit Care Med 185: 1096-1103.
- Nagakumar P, Hilliard T (2012) Recurrent lobar atelectasis in a child with cystic fibrosis. J R Soc Med 105: 50-52.
- Huisman C, de Graaff CS, Boersma WG (2002) Unilateral air bronchogram in a patient with cystic fibrosis. Chest 121: 1343-1344.
- McLaughlin AM, McGrath E, Barry R, et al. (2008) Treatment of lobar atelectasis with bronchoscopically administered recombinant human deoxyribonuclease in cystic fibrosis? Clin Respir J 2: 123-126.
- De Boeck K, Amaral MD (2016) Progress in therapies for cystic fibrosis. Lancet Respir Med 4: 662-674.
- Di Sant'Agnese PA (1953) Bronchial obstruction with lobar atelectasis and emphysema in cystic fibrosis of the pancreas. Pediatrics 12: 178-190.
- Cogen J, Emerson J, Sanders DB, et al. (2015) Risk factors for lung function decline in a large cohort of young cystic fibrosis patients. Pediatr Pulmonol 50: 763-770.
- Heltshe SL, Goss CH, Thompson V, et al. (2016) Short-term and long-term response to pulmonary exacerbation treatment in cystic fibrosis. Thorax 71: 223-229.
- Egan ME (2016) Genetics of cystic fibrosis: Clinical implications. Clin Chest Med 37: 9-16.
- Harness-Brumley CL, Elliott AC, Rosenbluth DB, et al. (2014) Gender differences in outcomes of patients with cystic fibrosis. J Womens Health (Larchmt) 23: 1012-1020.
- Cakir E, Uyan ZS, Ersu RH, et al. (2006) Mucoid impaction: An unusual form of allergic bronchopulmonary aspergillosis in a patient with cystic fibrosis. Pediatr Pulmonol 41: 1103-1107.
- Whitaker P, Brownlee K, Lee T, et al. (2011) Sequential bronchoscopy in the management of lobar atelectasis secondary to allergic bronchopulmonary aspergillosis. J Bronchology Interv Pulmonol 18: 57-60.
- Mateos Corral D, Cutz E, Solomon M, et al. (2009) Plastic bronchitis as an unusual cause of mucus plugging in cystic fibrosis. Pediatr Pulmonol 44: 939-940.
Corresponding Author
Concepción Prados-Sánchez, University Hospital La Paz, Cystic Fibrosis Unit, Pulmonology Service, Paseo Castellana 261, 28046 Madrid, Spain.
Copyright
© 2018 Prados-Sánchez C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.