Analyzing the Progress and Prevalence of Schedule II Opioid Prescription to Orthopedic Patients
Abstract
Background: In 2010, the Ambulatory Care Drug Database System (ACDD) reported that acetaminophen with hydrocodone topped the list of the three most prescribed medications across 49% of orthopedic facilities. Despite regulatory efforts radiated from the federal level, opioid prescription and addiction continues to exist among orthopedic patients.
Methods: We aimed at analyzing the trends of prescribing controlled substances to orthopedic patients post-2010 by querying the ACDD in the 2015 fiscal year, a median audit cutoff for the previous decade, and the National Health Interview Survey (NHIS) Questionnaire responses from 2011 to 2019. We used a primary drug query in the ACDD along with secondary therapeutic categorical filtration for preliminary data. After selecting the most cited qualifier, we tracked responses to a codified opioid usage per orthopedic care question for 9 years across all NHIS Questionnaires and concluded on prescription trends.
Results: With a mention rate of 44.94% and a mention count of 5,067,505, d03075, hydrocodone (generic) appeared as the third most popular schedule II opioid prescription, lowering the trend of popularity since 2010. The CDC did not develop a specific opioid-related orthopedic management question till the 2019 NHIS survey update. As a result, we could not use a z-test to compare between the first and second half of the decade to show there was significant difference between the prescription trends in the first half of the previous decade and the second half.
Conclusions: From 2011 to 2019, hydrocodone prescription rate decreased, and overall opioid prescription count was lower. Schedule II opioid options were replaced with a non-narcotic genre of pharmaceuticals. The increased surveillance on opioid prescription and awareness campaigns contributed to changes in trend; we predict the mention rate to fall further in the current decade.
Keywords
Oxycodone, Analgesics, Opioid, Postoperative period, Behavior, Addictive (Source: MeSH-NLM)
Background
Postoperative orthopedic patients are prone to develop moderate to severe pain, both acute and chronic. Schedule II opioids act as painkillers in acute pain management of postoperative orthopedic patients. In 2010, the Ambulatory Care Drug Database System (ACDD) reported that acetaminophen with hydrocodone topped the list of the three most prescribed medications across 49% of orthopedic facilities [1]. These narcotic drugs anchor to opioid receptors, a group of receptor proteins, on cerebral neurons, spinal cord, gut, and various body parts [2-8].
The binding between the receptor and the opioid particles leads to the blockage of pain messages sent from the peripheral nervous system (PNS) to the central nervous system (CNS) [9-13]. In addition to pain relief, these drugs also stimulate release of neurotransmitters responsible for feeling of euphoria (commonly known as a "high"). With chronic use, continuous increase in doses is needed to maintain analgesic effect as well as euphoria increasing the risk of dependence. Pain relief, however, is a relaxing feeling that may become an addiction if used for chronic pain management over a long period [14-22]. Despite regulatory efforts via federal and state authorities, opioid prescription remains irreplaceable [23-39]. While orthopedic surgeons agree on the effectiveness of narcotics for pain relief post-surgery, they are also concerned about its potential for abuse and dependence.
Despite continuing opioid abuse, we hypothesized that such abuse is no longer widespread in the system. Opioid prescription screening is now systematic, and the doses are now accounted. The US Department of Health and Human Services established the Prescription Drug Monitoring Programs (PDMPs), an electronic database that records the allocation of all controlled substance prescriptions. Even though such programs have existed for over a century, 70% of the modern, electronic programs were established in the first 15 years of the 21st century [40]. Authorized personnel may audit the dispense and dosage features to track down provider and patient transactions. Officials can track down patients who are receiving opioids from different sources. The application helps controllers measure the overall sum of opioids prescribed every day (in MME/day) and classify patients who are prescribed other drugs such as benzodiazepines that could raise dangerous, unsafe health risks, potentially even death. For such reasons, we expected to see a drop in the schedule II opioid prescription rate from 2010. Our initial focus was hydrocodone as it topped the list of prescription popularity in orthopedic facilities the previous decade. We also predicted that the second half of the previous decade would pose lesser dependence on opioid prescriptions. This study aimed to analyze the efficacy of opioid awareness program development at a systemic level from the point of view of orthopedic facilities.
Methods
This study was a retrospective query of governmentally released data on schedule II narcotics and survey responses to pain management for patients healing from fracture traumas across an adult population (ages 18 and above). We set a healing period for post-operative care within three months of a procedure. This study did not require IRB approval since the data came from public use databases for our independent study; a de-identified, public-use dataset that is freely accessible does not demand any organization to enter a formal written arrangement with the data provider. The ACDD was updated to reflect drugs in the database as of the 2015 survey year, which acts as a median cutoff point between 2010 and recent times. We also used the NHIS Questionnaire responses from 2012 to 2019. We used a primary drug query in the ACDD along with secondary therapeutic categorical filtration for preliminary data. After selecting the most cited qualifier, we tracked responses to a codified opioid usage question (OPD.0030.00.X) for eight years across all NHIS Questionnaires for adult (ages 18 years and above) samples.
We sensitized the consideration for narcotic group selection because we had to keep consistency to the comparative group from 2010. We also selected a specific survey question to make our data comparable, relatable, and reliable. Across numerous questions, the question that would indicate involvement of orthopedic facilities will involve fractures or broken bones. Only question no. OPD.0030.00.X mentioned a fracture and correlated it with opioid usage; even though there were many other questions about pain management or joint pain, tangential questions may involve non-orthopedic care such as pain medicine interventions, which would deter our aims. We aimed to perform a z-test between survey responses from 2011 to 2014 and from 2016 to 2019. Our participant number was in the tens of thousands; the z-test would summarize and indicate whether the two population means (i.e., responders who said yes to taking opioids for pain management) are different.
Results
Schedule II opioids
There was a total of 122 resulting groups of prescription drugs. These groups were a single entity in composition status, and all were DEA Schedule II. With a citation count of 9,832,801, oxycodone appeared as the most prescribed schedule II opioid (Table 1). Hydrocodone, though on top 3, was superseded by morphine with a numerical difference of 2,921,631 citations. It is important to remember that most drugs within the 122 results had brand name repetitions, which required us to hold a cumulative comparison (Table 2). Hydrocodone, for example, had five brand names under its generic name, each with different citation counts that may contribute to its cumulative citations. Even though the top four generic drugs remain the same in a cumulative comparison, methylphenidate takes precedence over hydromorphone as the fifth most prescribed generic drug.
Opioid prescription to orthopedic patients
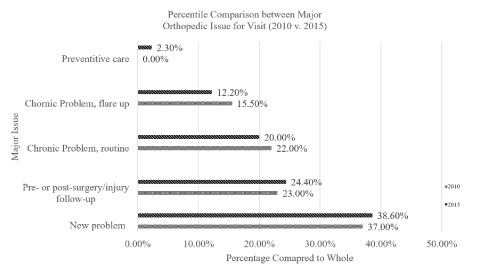
According to the ACDD, postoperative care-receivers represented 20.8% of the orthopedic patients, second to patients with new problems (Figure 1). The CDC failed to incorporate a question specific to fracture care and prescription medications on the surveys from 2011 to 2017. Even though it asked the responders questions about fractures and medications, they did not correlate across any of the surveys. One improvement was noted in the 2018 survey that inquired patients about acute pain management with a prescription opioid; 65.50% of the responders responded "yes." The CDC redesigned its surveys from 2019; according to the agency, "In 2019, for the first time in over 20 years, the NHIS questionnaire was redesigned to increase relevance, enhance data quality, and minimize respondent burden.
The wording of some questions and answer choices was revised to improve the quality of measurement. Some questions were dropped, and others were added, changing the context within which some questions were asked [41]." The surveys from 2019 onwards incorporated a question for patients to report prescription opioid dispensation for fracture management. We noted a minimal drop of 1.16% between acute pain management using opioids from 2018 to 2019 (Table 3). The code books for data release are under construction for the 2020 and 2021 fiscal years; we purposefully left out 2020 from this study due to the appearance of COVID-19. The NAIHS became an over-the-phone survey due to the pandemic, which drastically changed its participant frequency; 2020 can be a notable outlier. We could not perform our desired z-test since CDC datasets failed to specify opioid usage questionnaires to fracture management until the end of the decade.
Discussion
The opioid crisis ethically challenged the medical community, particularly orthopedic surgeons since they often deal with such narcotic drugs in postoperative care. At the brink of a new decade in February 2019, the American Academy of Orthopedic Surgeons (AAOS) audited various orthopedic facilities in a randomized, nationally distributed survey to check current orthopedic prescription trends [42]. The academy acquired data from 555 orthopedic facilities and concluded that most physicians do not prescribe refills on schedule II opioids and prescribe opioids to adults more often than teenagers. To cross-check and audit internally, orthopedic researchers in the Nemours system, who mainly treats children and adolescents, audited surgical charts on pediatric anterior cruciate ligament (ACL) surgery from 2013 to 2017 on August 2019 [43].
Even though pediatric procedures are not in the center of our study, this audit is crucial for capturing the progress of orthopedic surgery at large, especially since teenagers were a part of the AAOS study. The surgical charts indicated that in the joint nerve block audit, fewer than half of the patients (ages 12-17) had opioid administration intraoperatively or in the PACU. While both queries tend to show that orthopedic surgery overall lessened its reliance on opioids over the second decade of the 21st century, we aimed to design another way to confirm that trend. Even though the AAOS researchers targeted maintaining objectivity in their study at Raneses E et. al. [42] we must note that their method had design limitations. Five hundred fifty-five orthopedic facilities represent a fraction of orthopedic care centers around the United States. Furthermore, there is a possibility of response bias in such email surveys since orthopedic surgeons may have responded in a way that would tend to provide a more favorable outcome. In addition, the study conducted at the Nemours system may also have had an internal bias. We took note of these previous lacking so we can avoid such mistakes in our design, a reason why we relied on data collected by a third party. The CDC's conflict of interest with orthopedic opioid care is minimal to none. At the same time, all NHIS surveys may have the risk of reporting bias about patient medications; however, these anonymous surveys are more likely to be objective than biased due to minimal incentivization among the sample population. In addition, NHIS survey samples are quantitatively higher than other related studies, posing more diversity in our study.
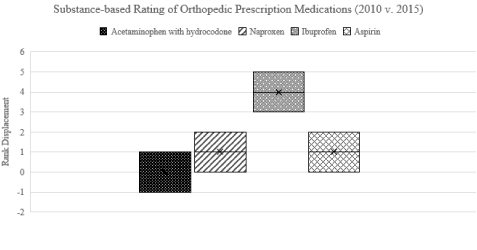
We first focused on prescription trends and then panned our focus to patient reports. As the data from ACDD shows, hydrocodone usage significantly dropped over the previous decade (displacement of 2 ranks) (Table 3). The NAMCS database to access the Orthopedic Surgery Fact Sheet released in 2015 cross-checks this indication [44]. We observed that ibuprofen and aspirin prevailed over hydrocodone prescription in this report (Figure 2). Ibuprofen is a nonsteroidal drug with no DEA control schedule, and, likewise, aspirin has no DEA control schedule, thus showing that orthopedic surgeons have moved away from overprescribing schedule II opioids, and rather relying more on non-narcotics-based care for pain control. While our cumulative data collection shows the continued persistence of controlled substance prescriptions, we conclude that such data trend applies more to general medicine than orthopedic surgery which has reduced reliance on narcotics.
We then focused on confirming this trend with patient-backed data. Unfortunately, NHIS surveys overlooked opioid usage in orthopedic care specifically until 2018-2019. Across all surveys from 2011 to 2017, the surveys included trauma reporting questions, such as Question AHS.304-01.000 from the 2013 NHIS Questionnaire, which asked, "How long have you had a fracture, bone, or joint injury? [45]" While such questions contribute to general orthopedic knowledge, they fail to pinpoint prescription culture within the orthopedic community. Even though we could not perform a z-test to compare opioid prescription frequency between two halves of the previous decade, we were able to compare two years of datasets conducted among patients between 2018 and 2019 questionnaires, which demonstrated a 1.16% drop in schedule II prescription frequency in our highly quantified sample (p = 2,926). While this may be too early to conclude on a progressive trend, it is an indication that orthopedic researchers will be able to objectively compare prescription frequencies in the upcoming decade since the NHIS questionnaires for 2020 and 2021 both include OPD.0030.00.X in its question sample. Based on preliminary data and the Orthopedic Surgery Fact Sheet, our research demonstrates non-controlled oral analgesic agents had preceded prescription frequency over controlled medications during the past 2010s decade, indicating the trend away from controlled substances as once heavily prescribed in 2010 and earlier.
Acknowledgment
N/A.
Financing
N/A.
Conflict of Interest Statement by Authors
N/A.
Compliance with Ethical Standards
The entirety of this independent research consists of "public-use" de-identified data sets that do not require IRB approval, as the data from such sources is made public.
References
- Centers for Disease Control and Prevention (CDC) (2010) NAMCS fact sheet for orthopedic surgery.
- Stoeber M, Jullie D, Lobingier BT, et al. (2018) A genetically encoded biosensor reveals location bias of opioid drug action. Neuron 98: 963-976.
- Arttamangkul S, Alvarez Maubecin V, Thomas G, et al. (2000) Binding and internalization of fluorescent opioid peptide conjugates in living cells. Mol Pharmacol 58: 1570-1580.
- Calebiro D, Nikolaev VO, Gagliani MC, et al. (2009) Persistent cAMP-signals triggered by internalized g-protein-coupled receptors. PLoS Biol 7: e1000172.
- Calebiro D, Nikolaev VO, Persani L, et al. (2010) Signaling by internalized gprotein-coupled receptors. Trends Pharmacol Sci 31: 221-228.
- Grady EF, Garland AM, Gamp PD, et al. (1995) Delineation of the endocytic pathway of substance P and its seven-transmembrane domain NK1 receptor. Mol Biol Cell 6: 509-524.
- Henry AG, Hislop JN, Grove J, et al. (2012) Regulation of endocytic clathrin dynamics by cargo ubiquitination. Dev Cell 23: 519-532.
- Horton AC, Ehlers MD (2003) Dual modes of endoplasmic reticulum-to-Golgi transport in dendrites revealed by live-cell imaging. J Neurosci 23: 6188-6199.
- Huang W, Manglik A, Venkatakrishnan AJ, et al. (2015) Structural insights into µ-opioid receptor activation. Nature 524: 315-321.
- Autry AE, Adachi M, Nosyreva E, et al. (2011) NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 475: 91-95.
- Ballard ED, Ionescu DF, Vande Voort JL, et al. (2014) Improvement in suicidal ideation after ketamine infusion: relationship to reductions in depression and anxiety. J Psychiatr Res 58: 161-166.
- Becker DE, Reed KL (2012) Local anesthetics: Review of pharmacological considerations. Anesth Prog 59: 90-101, quiz 102-103.
- Bourke DL, Malit LA, Smith TC (1987) Respiratory interactions of ketamine and morphine. Anesthesiology 66: 153-156.
- Ballard ED, Ionescu DF, Vande Voort JL, et al. (2014) Improvement in suicidal ideation after ketamine infusion: Relationship to reductions in depression and anxiety. J Psychiatr Res 58: 161-166.
- Ballard ED, Wills K, Lally N, et al. (2017) Anhedonia as a clinical correlate of suicidal thoughts in clinical ketamine trials. J Affect Disord 218: 195-200.
- Bobo WV, Miller SC (2002) Ketamine as a preferred substance of abuse. Am J Addict 11: 332-334.
- Bonanno FG (2002) Ketamine in war/tropical surgery (a final tribute to the racemic mixture). Injury 33: 323-327.
- Bresink I, Danysz W, Parsons CG, et al. (1995) Different binding affinities of NMDA receptor channel blockers in various brain regions-indication of NMDA receptor heterogeneity. Neuropharmacology 34: 533-540.
- Can A, Zanos P, Moaddel R, et al. (2016) Effects of ketamine and ketamine metabolites on evoked striatal dopamine release, dopamine receptors, and monoamine transporters. J Pharmacol Exp Ther 359: 159-170.
- Debelak K, Morrone WR, Grady O KE, et al. (2013) Buprenorphine + naloxone in the treatment of opioid dependence during pregnancy: Initial patient care and outcome data. Am J Addict 22: 252-254.
- Deck D, Wiitala W, McFarland B, et al. (2009) Medicaid coverage, methadone maintenance, and felony arrests: Outcomes of opiate treatment in two states. J Addict Dis 28: 89-102.
- Fatima M, Srivastav S, Mondal AC (2017) Prenatal stress and depression associated neuronal development in neonates. Int J Dev Neurosci 60: 1-7.
- CDC (2015) National center for health statistics. Multiple cause of death 1999-2014 on CDC WONDER online database, released 2015. Data are from the Multiple Cause of Death Files, 1999-2014, as compiled from data provided by the 57 vital statistics jurisdictions through the Vital Statistics Cooperative Program.
- CDC (2014) Quickstats: Rates of drug poisoning deaths involving heroin, by selected age and racial/ethnic groups-United States, 2002 and 2011. Morbidity and Mortality Weekly Report 63: 595.
- CDC (2017b) QuickStats: Rates of drug overdose deaths involving heroin, by selected age groups-United States, 2006-2015. Morbidity and Mortality Weekly Report 65: 1497.
- Cerdá M, Ransome Y, Keyes KM, et al. (2013) Prescription opioid mortality trends in New York City, 1990-2006: Examining the emergence of an epidemic. Drug Alcohol Depend 132: 53-62.
- Cicero TJ, Ellis MS, Surratt HL, et al. (2014) The changing face of heroin use in the United States: A retrospective analysis of the past 50 years. JAMA Psychiatry 71: 821-826.
- Compton WM, Jones CM, Baldwin GT (2016) Relationship between nonmedical prescription-opioid use and heroin use. N Engl J Med 374: 154-163.
- Coplan PM, Sessler NE, Harikrishnan V, et al. (2017) Comparison of abuse, suspected suicidal intent, and fatalities related to the 7-day buprenorphine transdermal patch versus other opioid analgesics in the national poison data system. Postgrad Med 129: 55-61.
- Dart RC, Surratt HL, Cicero TJ, et al. (2015) Trends in opioid analgesic abuse and mortality in the United States. N Engl J Med 372: 241-248.
- Davis AM, Inturrisi CE (1999) D-Methadone blocks morphine tolerance and N-methyl-d-aspartate-induced hyperalgesia. J Pharmacol Exp Ther 289: 1048-1053.
- Davis M, Goforth HW, Gamier P (2013) Oxycodone combined with opioid receptor antagonists: Efficacy and safety. Expert Opin Drug Saf 12:389-402.
- Hughes A, Williams MR, Lipari RN, et al. (2016) Prescription drug use and misuse in the United States: Results from the 2015 national survey on drug use and health. Ann Intern Med 167: 293-301.
- IOM (2011) Relieving pain in America: A blueprint for transforming prevention, care, education, and research. Washington, DC: National Academy Press.
- Jones CM (2013a) Heroin use and heroin use risk behaviors among nonmedical users of prescription opioid pain relievers-United States, 2002-2004 and 2008-2010. Drug Alcohol Dependence 132: 95-100.
- Jones CM, Lurie PG, Compton WM (2016) Increase in naloxone prescriptions dispensed in U.S. retail pharmacies since 2013. Am J Public Health 106: 689-690.
- Jones HE, Kaltenbach K, Heil SH, et al. (2010) Neonatal abstinence syndrome after methadone or buprenorphine exposure. N Engl J Med 363: 2320-2331.
- Lankenau SE, Schrager SM, Silva K, et al. (2012) Misuse of prescription and illicit drugs among high-risk young adults in los angeles and New York. Public Health Res 1: 22-30.
- Wheeler E, Jones T, Gilbert M, et al. (2015) Opioid overdose prevention programs providing naloxone to laypersons-United States, 2014. Morbidity and Mortality Weekly Report 64: 631-635.
- Marilyn Bulloch (2021) The Evolution of the PDMP. Pharmacy Times.
- Centers for Disease Control and Prevention (CDC) (2019) Preliminary evaluation of the impact of the 2019 national health interview survey questionnaire redesign and weighting adjustments on early release program estimates.
- Raneses E, Secrist ES, Freedman KB, et al. (2019) Opioid prescribing practices of orthopaedic surgeons: Results of a national survey. J Am Acad Orthop Surg 27: e166-e172.
- Santana L, Lovejoy JF, Kiebzak G, et al. (2019) Comparison of pain scores and medication usage between three pain control strategies for pediatric anterior cruciate ligament surgery. Cureus 11: e5498.
- Centers for Disease Control and Prevention (CDC) (2015) Orthopedic surgery fact sheet from the national ambulatory medical care survey.
- Centers for Disease Control and Prevention (CDC) (2013) NHIS Questionnaire - Sample Adult Identification.
Corresponding Author
Ahmed Nahian, California Baptist University Lake Erie College of Osteopathic Medicine, 8432 Magnolia Ave CMB 2682, Riverside, CA 92504, USA, Tel: 347-241-2252.
Copyright
© 2021 Nahian A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.