Temporal Variation of Nitrogen and Phosphorus Species Distribution on Total Phytoplankton Biomass in Eutrophic Lakes
Abstract
To aid in the eutrophication management, the relationships between total algal biomass in response to varying nitrogen and phosphorus species concentrations, Volatile (VSS), and Total Suspended (TSS) Solids in the water column were investigated. Sampling was conducted at the deepest portion of the reservoir for analysis of phosphorus, nitrogen, chlorophyll a, VSS, and TSS. The study showed both VSS and TSS distribution coincided with the vertical distribution of chlorophyll a. The chlorophyll a was positively and significantly correlated to TSS and VSS. Correlation analysis revealed that the VSS and TSS were significantly related to the phytoplankton growth. No linear relationship was observed between total phosphorus and chlorophyll a biomass and result indicated that the phytoplankton growth declined when nitrogen becomes the primary limiting nutrient after the phosphorus concentration of 200 µg/L in the water column. The soluble reactive phosphorus and dissolved inorganic nitrogen remained low throughout the sampling period; however, total dissolved phosphorus and dissolved organic nitrogen remained high and varied according to the phytoplankton growth. This study may provide a basis for further investigations of nutrient species and phytoplankton growth studies.
Keywords
Secchi depth, Total phosphorus, Total nitrogen, Dissolved organic nitrogen, Dissolved organic phosphorus
Introduction
Aquatic ecosystems worldwide experience cultural eutrophication accompanied by algal blooms that cause serious socioeconomic and ecological costs, which impact fisheries, drinking water, agriculture, tourism, and contribute to summer fish kills and prolong anoxia [1-3]. Many studies have reported that cultural eutrophication has influenced the harmful algal blooms [4-6] and those blooms were threatened the some of the world's largest and important freshwater bodies, including Lakes Erie (US-Canada); Taihu (China) and Victoria (Africa) [7-9]. Many genera of cyanobacteria are known to produce cyanobacterial toxins, including hepatotoxins and neurotoxins that cause the health risk for both animals and humans [10,11]. Increasing nutrient concentrations (nitrogen, N, and phosphorus, P) have been considered the key factors responsible for promoting cyanobacterial blooms, and they are predicted to increase with increased nutrient availability, especially phosphorus in eutrophic and hypereutrophic lakes [12]. Studies have reported that cyanobacteria population potentially dominated at total phosphorus concentrations of 100-1000 µg L-1 [13,14] and phosphorus availability is the key factor controlling cyanobacterial blooms [15].
Changes of total phytoplankton biomass (Chlorophyll a) has been widely used to assess the ecological status of the aquatic ecosystems because of their responses to prevailing nutrient conditions and different environmental conditions [16,17]. Understanding of the relationship between nutrient concentration and algal biomass is paramount in the efforts for effective eutrophication management. The relationship between N and P enrichment and increased algal biomass and productivity is well studied; nevertheless, the efforts for links between varying N and P partitioning and algal biomass in the eutrophic lakes are rarely made. Inorganic N (ammonium and nitrate) and P (orthophosphate) partitioning and their bioavailability have been paid much attention in the lake ecosystems; however, Dissolved Organic N (DON), Dissolved Organic P (DOP), Particulate N (PN), Particulate P (PP), and their role in the lake eutrophication is overlooked.
In the most aquatic ecosystems, the largest pool of nitrogen is associated with the dissolved organic [18,19] and studies have shown that the significant amount of DON contributes to the phytoplankton nutrition and the coastal eutrophication [19-21]. Herndon and Cochlan [22] reported that capability of utilizing DON by some harmful algal species; however, other phytoplankton groups such as diatoms and dinoflagellates showed to have an affinity for inorganic N [23-25]. However, little is known about the dynamics of organic N and P fractions and their bioavailability in the freshwater eutrophic ecosystems.
The present study was carried out during seasonal stratification period at a eutrophic reservoir used for recreational purposes. The aim of this study was to investigate the variations of total algal biomass (as chlorophyll a) in response to varying N and P species concentrations in the water column during the lake stratified period. Nitrogen and phosphorus species distribution with algal biomass was studied by measuring and calculating N and P species in the water column during bloom conditions. Information on N and P composition in the eutrophic lakes is important to understand its biogeochemical cycles and this study will help for further investigations of contributions of N and P fractions to eutrophication in lake ecosystems.
This reservoir is under the administration of the Department of Game and Fish in the State of North Dakota, and findings from this study may provide further information for them to improve the existing eutrophication management system (an existing artificial destratification system) in response to the temporal variations of the N and P fractions in the water column.
Materials and Methods
Study area
The research was conducted in the Heinrich-Martin Dam Impoundment (HMDI), which is a small and shallow impoundment located in the southeastern North Dakota, USA. The thermal stratification develops in the summer and the impoundment artificially aerated except in the months of July and September. The existing aeration system in the HMDI is an artificial destratification system placed in the deepest area (10 m) of the impoundment. A complete description of the existing aeration system is provided in Balangoda [26].
Sampling
Water samples were obtained with a nonmetallic Van Dorn vertical water sampler and the water quality profiles were collected at the deepest location on a weekly basis in June through the stratification period, until November in 2011. Samples were collected for the laboratory analysis of phosphorus and nitrogen components (nitrate-nitrogen, nitrite-nitrogen, ammonia nitrogen, total nitrogen, total dissolved nitrogen, soluble reactive phosphorus, acid hydrolyzable phosphorus, and total phosphorus), chlorophyll a, Volatile Suspended Solids (VSS), and Total Suspended Solids (TSS). In addition, total dissolved organic nitrogen, total dissolved inorganic nitrogen, total particulate nitrogen, total reactive phosphorus, total dissolved phosphorus, total organic phosphorus, and total particulate phosphorus have been calculated using measured nitrogen and phosphorus components in the water column. To determine the vertical variations of VSS and TSS, the water samples were collected from the different depths such as 0.5 m from the surface, Secchi depth, and near-bottom water (1.5 m from the bottom) at the deepest location.
Analytical methods
The VSS was measured after igniting the residue produced by TSS in a muffle furnace at a temperature of 550 °C. The VSS measurements were taken after a constant weight was obtained. Water samples for nitrogen, phosphorus, and chlorophyll a were analyzed according to the American Public Health Association (APHA) [27] standard methods. A description of the chemical analyses can be found in Balangoda [26], while chlorophyll a analysis is detailed in Balangoda, et al. [28].
Statistical analyses
The MINITAB 17.0 version was used for all the correlation and regression analyses. The significance of the results was determined at the 0.05 probability level (P < 0.05).
Results
Volatile Suspended Solids (VSS) - chlorophyll a relationship
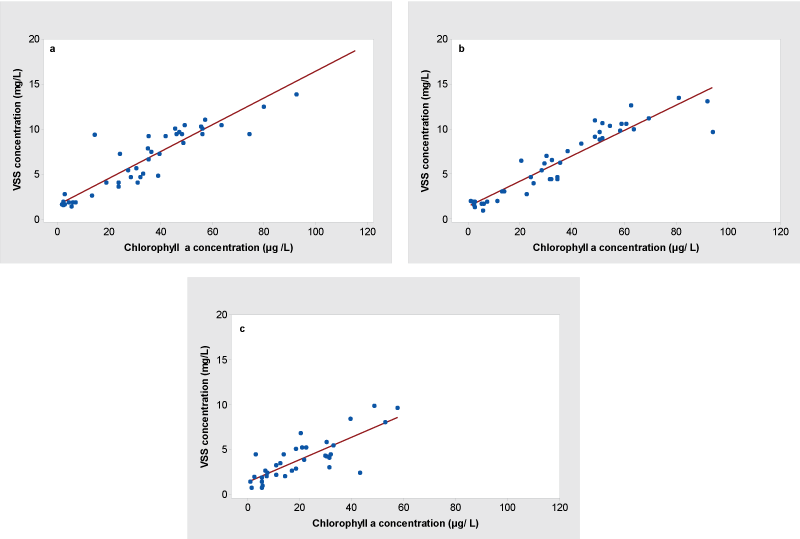
Chlorophyll a (Chl a) as a photosynthetic pigment represents all type of phytoplankton is a measurable parameter for the total production of phytoplankton growth. The strong correlations between Chl a and the VSS were observed at the surface (r = 0.928) and the Secchi depth level (r = 0.934) compared to the near-bottom water (0.802). The VSS/Chl a ratio and the strength of correlation varied among the depths. The linear regression analysis showed a significant correlations between Chl a and the VSS at the surface (R2 = 0.8617, P < 0.05) (Figure 1a) and the Secchi depth (R2 = 0.8729, P < 0.05) (Figure 1b) compared to the near-bottom water (R2 = 0.6432, P < 0.05) (Figure 1c). The highest VSS/Chl a ratio was observed at the Secchi depth level followed by the surface and the near-bottom water, indicated that a source of the most of the VSS was phytoplankton at both the surface and the Secchi depth levels than the near-bottom water. The VSS/Chl a ratios showed variations of algal biomass along the depths and contribution of phytoplankton biomass to total organic matter content at each depth.
Volatile suspended solids - Total Suspended Solids (TSS) relationship
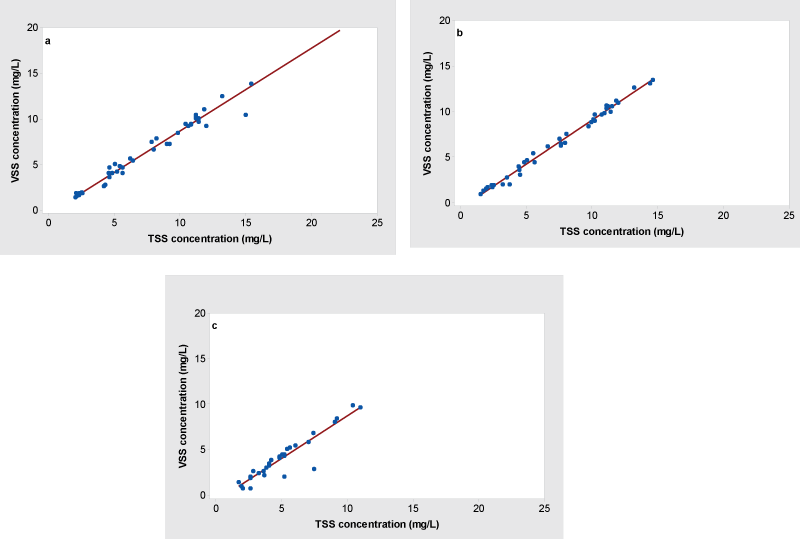
Total suspended solids measure solids materials, including both inorganic and organic particles suspended in the water which will not pass through a filter. Strong correlations between TSS and VSS were observed at the surface (r = 0.987) and the Secchi depth level (r = 0.995) compared to the near-bottom water (0.941). The VSS/TSS ratio and strength of the correlation varied along the depths (Figure 2). The linear regression analysis showed significant correlations between TSS and VSS at the surface (R2 = 0.9745, P < 0.05) (Figure 2a) and the Secchi depth (R2 = 0.9890, P < 0.05) (Figure 2b) compared to the near-bottom water (R2 = 0.8852, P < 0.05) (Figure 2c). The highest VSS/TSS ratio was observed at the surface followed by the Secchi depth level, and the near-bottom water. The VSS/TSS ratios indicated that the surface water column had the highest amount of organic matter in its suspended solids which most likely comprised of phytoplankton.
Time series analyses of phosphorus species
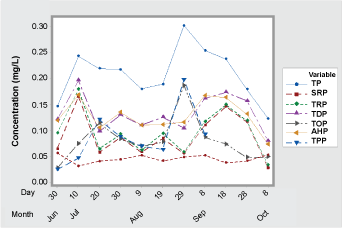
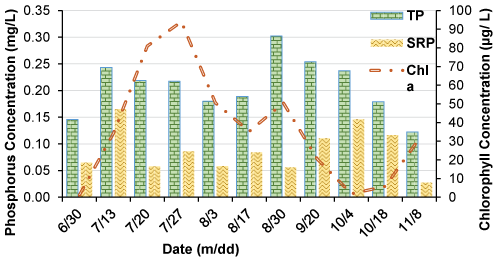
Time series analyses of phosphorus species such as Total Phosphorus (TP), Soluble Reactive Phosphorus (SRP), Total Reactive Phosphorus (TRP), Total Dissolved Phosphorus (TDP), Total Organic Phosphorus (TOP), Acid Hydrolyzable Phosphorus (AHP), and Total Particulate Phosphorus (TPP) are shown in Figure 3. The TP concentration represented the sum of all the available forms of phosphorus species in the water column. The concentrations of phosphorus species (except the SRP concentration) increased by July 10th when an aeration system was turned off and the impoundment started to stratify for the season. The SRP which is the readily bioavailable form of phosphorus remained low among other phosphorus species in the water column. The TP concentration fluctuated in response to other phosphorus species in the water column during the stratified period (July 10th - Sep 1st). The TDP and AHP were followed closer to the variations of TP and TDP and the second most available forms of phosphorus species in the water column. The second peak concentration of TP was observed by Sep 1st in response to the start of the aeration system. The TP concentration was gradually decreased by the end of the sampling of Oct 8th when the other phosphorus species (TRP, TDP, TOP, AHP, and TPP) continued to decrease in the water column.
Time series analyses of nitrogen species
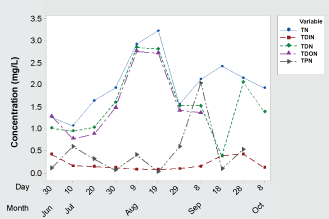
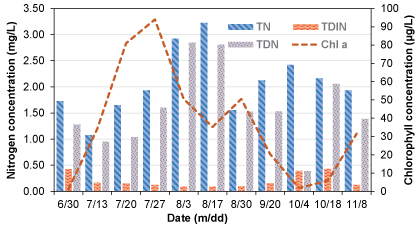
Time series analyses of nitrogen species such as Total Nitrogen (TN), Total Dissolved Inorganic Nitrogen (TDIN), Total Dissolved Nitrogen (TDN), Total Dissolved Organic Nitrogen (TDON), and Total Particulate Nitrogen (TPN) are shown in Figure 4. The TN is composed of both total organic and total inorganic nitrogen. The total organic nitrogen is comprised of both algae and sediments while the total inorganic nitrogen is comprised of ammonia nitrogen (NH4-N), nitrate-nitrogen (NO3-N), and nitrite-nitrogen (NO2-N). The TN concentration was between 1 mg/L and 1.5 mg/L by the end of the June, and TN concentration has reached a peak during the lake stratified period. The TN concentration fluctuated when aeration was turned on by Sep 1st due to a mixed condition that led to resuspend the organic detritus as well as enhanced the organic matter decomposition due to increased dissolved oxygen concentration by an aeration system at the bottom layer (Figure 4).
The TDON concentration was calculated using values from TDN (Total Dissolved Nitrogen) and TDIN (Total Dissolved Inorganic Nitrogen: NH4-N + NO3-N + NO2-N); TDON = (TDN - TDIN). The source of the TDON was most likely microbial decomposition of particulate organic nitrogen (phytoplankton, zooplankton, and detritus) and not directly bioavailable for algal growth. The TDON concentration was varied closer to the TN concentration changes during the study period (Figure 4). The TPN is calculated using values from TN and TDN; TPN = (TN -TDN), and TPN is most likely consisted of algae, zooplankton, and other organic detritus.
Inorganic nutrients are up taken by phytoplankton for their growth. The TDIN (Total Dissolved Inorganic Nitrogen) is the available part of nitrogen for algal growth. The TDIN concentration was 0.4 mg/L and gradually decreased to 0.09 mg/L during the stratified period and again started to increase when the aeration system was turned on by Sep 1st which allowed nutrient to mix along the depths.
Correlation between chlorophyll a with TP and SRP
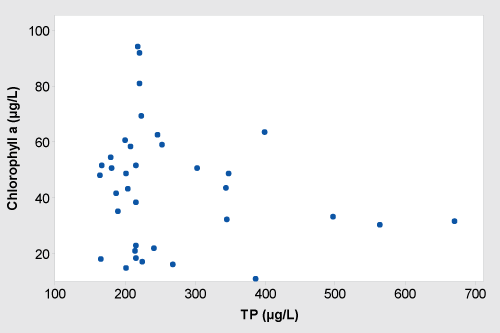
The correlation between TP and chlorophyll a is shown in Figure 5. Chlorophyll a concentration reached its maximum concentration (90 µg/L) when TP concentration was approximately 200 µg/L. There was no linear relationship observed between TP and Chl a (Figure 5). Chlorophyll a concentration dropped even TP was in its maximum concentration of 690 µg/L in the water column.
The Figure 6 showed temporal variation of TP and SRP with Chl a through the sampling period. Chl a concentration was gradually increased and reached a peak value of approximately 90 µg/L during the month of July; however, Chl a concentration declined steadily from August to October with the water temperature changes. The SRP concentration remained low throughout the study period and continued to be low when chlorophyll a concentration reached its maxima. The TP concentration partially represented by phytoplankton (chlorophyll a) biomass as organic phosphorus. As shown in Figure 6, Chlorophyll a concentration increased with increasing TP concentration; however, TP concentration continued to increase even Chl a concentration completely declined most likely due to an increase of organic biomass as dead phytoplankton biomass. The Figure 6 indicates that the contribution of phytoplankton biomass as Chl a to the internal cycling of phosphorus in the water column.
Temporal variation of nitrogen species (TN, TDIN, and TDN) and chlorophyll a
Temporal variation of TN, TDIN, and TDN with Chlorophyll a is shown in Figure 7. The TN concentration represented the sum of the nitrogen present in the water column; however, the bioavailable form of nitrogen for plants and algal growth is TDIN. Similar to SRP (Figure 6), the TDIN also remained low throughout the sampling period; however, in comparison with TN concentration, the second most available nitrogen species in the water column was TDN. The TDN is the sum of dissolved organic and inorganic nitrogen in the water column and results indicated that the majority of TDN was the form of dissolved organic nitrogen in the water column. The variation of TDN closely followed with the temporal variation of TN and remained high in the water column than TDIN concentration.
Discussion
The suspended solids (VSS and TSS) and Chl a showed strong correlations along the depths in the water column. Studies have reported that similar findings for correlation between suspended solids and Chl a [29]. The VSS/Chl a ratios may have an influence on the presence of an amount of organic matter content at the each depth other than the phytoplankton cells. Findings also indicated that the most of the Chl a reported at the epilimnion (both surface and Secchi depth level) than the hypolimnion (near-bottom water) was live organic matter (as live phytoplankton) than the organic matter resuspension from the sediment during the lake stratified period.
The use of chlorophyll to estimate the algal biomass in aquatic habitats is crucial when biovolume varies among species and environmental conditions [30,31]. Summer chlorophyll a concentration and measured TP concentration at the HMDI did not show the linear relationship as predicted by Dillon and Rigler [32]. However, studies have shown that chlorophyll-phosphorous relationships can be changed due to several lake features such as different grazing pressure of zooplankton on the algae, presence or absence of abiotic turbidity or other variables [33-35]. Scheffer [36] reported that another reason that data doesn't fit chlorophyll-nutrient model is if the data comes from the shallow polymictic lake like HMDI. Chlorophyll a content at such water bodies often depend upon photic conditions and zooplankton grazing pressure than nutrient concentration in the water column. Kufel [37,38] has shown that the lack of correlation between chlorophyll a and nutrient in shallow lakes while McCauley, et al. [39] showed that nitrogen limitation could also be one of the attributes that change the relationship between Chl a-TP. Studies have shown that TP-Chl a relationship is actually sigmoidal and Chl a response is weak when TP concentration is both high ( > 100 µg/L) and low (< 10 µg/L) concentrations [34,40].
Dissolved Inorganic Phosphorus (SRP) and Dissolved Inorganic Nitrogen (TDIN) typically depleted during the vegetative period. Chlorophyll a biomass will decline if dissolved inorganic nutrients are not resupplied to the surface water column from the nutrient rich near-bottom layers during the lake stratified period. The results indicated that among phosphorus species TDP was the second most available phosphorus in the water column and most of the TDP was in the form of Dissolved Organic Phosphorus (DOP). The most of the nitrogen was in the form of Dissolved Organic Nitrogen (DON), and previous studies have shown the bioavailability of DOP and DON [41-43]. The findings indicated that declined of phytoplankton growth most likely due to the limited supply of dissolved inorganic nitrogen and phosphorus and unavailability to the uptake of those DOP and DON.
The high DON and DOP were observed in the near surface water column which implies that DON and DOP produced by the phytoplankton in the epilimnion, and both DON and DOP were as a part of the euphotic zone processes. The observed decrease of TN, TDN, and DON concentrations in the surface water column caused by mixing of the water column due to restarting of the existing aeration system and water column was mixed. Shallow, polymictic eutrophic lakes can display complex nutrient dynamics due to wind drove nutrient resuspension.
Conclusions
The study shows that both VSS and TSS distribution coincided with the vertical distribution of Chl a in the water column. Regression analyses indicated that the Chl a was significantly correlated with the TSS and VSS. No linear relationship was observed between TP and Chl a in the water column. The SRP and TDIN concentrations remained low, and TDP and TDN remained the second most available form of phosphorus and nitrogen, respectively. Moreover, the results indicated that those TDP and TDN were not bioavailable for the cyanobacterial bloom (Aphanizomenon flos-aquae) reported in the HMDI during summer stratified period.
Acknowledgements
This research was supported by the North Dakota Water Resources Research Institute fellowship. I thank North Dakota Game and Fish Department and Department of Civil and Environmental Engineering for providing facilities for this research; Shafiqur Rahman and G. Padmanabhan for their guidance; Malcolm Butler for providing necessary water quality samplers; Wei Lin and Vesselina Volkov for assisting in field sampling.
References
- Paerl HW (2008) Nutrient and other environmental controls of harmful cyanobacterial blooms along the freshwater - marine continuum. Adv Exp Med Biol 619: 217-237.
- Watson SB, Whitton BA, Higgins SA, et al. (2015) Harmful algal blooms. Freshwater Algae of North America. Academic Press, San Diego, CA, USA, 873-920.
- Carmichael WW, Boyer GL (2016) Health impacts from cyanobacteria harmful algae blooms: Implications for the North American Great Lakes. Harmful Algae 54: 194-212.
- Glibert PM, Anderson DA, Gentien P, et al. (2005) The global, complex phenomena of harmful algal blooms. Oceanography 18: 136-147.
- Glibert PM, Burkholder JM (2006) The complex relationships between increasing fertilization of the Earth, coastal eutrophication, and Proliferation of Harmful Algal Blooms. Ecology of Harmful Algae 341-354.
- Heisler JP, Gilbert J, Burkholder J, et al. (2008) Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 8: 3-13.
- Havens KE, Fukushima T, Xie P, et al. (2001) Nutrient dynamics and the eutrophication of shallow lakes Kasumigaura (Japan), Donghu (PR China), and Okeechobee (USA). Environmental Pollution 111: 263-272.
- Qin B, Zhu G, Gao G, et al. (2010) A drinking water crisis in Lake Taihu, China: linkage to climatic variability and lake management. Environ Manage 45: 105-112.
- Abell JM, Ozkundakci D, Hamilton DP, et al. (2012) Latitudinal variation in nutrient stoichiometry and chlorophyll- nutrient relationships in lakes: A global study. Applied Limnology 181: 1-14.
- Klisch M, Haeder DP (2008) Mycosporine-like amino acids and marine toxins-the common and the different. Mar Drugs 6: 147-163.
- Sivonen K, Borner T (2008) Bioactive compounds produced by cyanobacteria. The Cyanobacteria Molecular Biology, Genomics and Evolution. Caister Academic Press, 159-198.
- Rigosi A, Cayelan CC, Ibelings BW, et al. (2014) The interaction between climate warming and eutrophication to promote cyanobacteria is dependent on trophic state and varies among taxa. Limnology and Oceanography 59: 99-114.
- Watson SB, McCauley E, Downing JA (1997) Patterns in phytoplankton taxonomic composition across temperate lakes of differing nutrient status. Limnology and Oceanography 42: 487-495.
- Downing JA, Watson SB, McCauley E (2001) Predicting cyanobacteria dominance in lakes. Can J Fish Aquat Sci 58: 1905-1908.
- Schindler DW, Hecky RE, Findlay DL, et al. (2008) Eutrophication of lakes cannot be controlled by reducing nitrogen input: results of a 37 year whole ecosystem experiment. Proc Natl Acad Sci U S A 105: 11254-11258.
- Reynolds CS (2000) Phytoplankton designer-or how to predict compositional responses to trophic-state change. Hydrobiologia 424: 123-132.
- Yerli SV, Kıvrak E, Gurbuz H, et al. (2012) Phytoplankton community, nutrients and Chlorophyll a in Lake Mogan (Turkey); with comparison between current and old data. Turkish Journal of Fisheries and Aquatic Sciences 12: 95-104.
- Berman T, Chava S (1999) Algal growth on organic compounds as nitrogen sources. J Plankton Research 21: 1423-1437.
- Bronk DA, See JH, Bradley P, et al. (2007) DON as a source of bioavailable nitrogen for phytoplankton. Biogeosciences 4: 283-296.
- Bronk DA (2002) Dynamics of DON in biogeochemistry of marine dissolved organic matter. Academic Press, San Diego, 153-249.
- Berman T, Bronk DA (2003) Dissolved organic nitrogen: A dynamic participant in aquatic ecosystems. Inter-Research Aquatic Microbial Ecology 31: 279-305.
- Herndon J, Cochlan WP (2007) Nitrogen utilization by the Raphidophyte Heterosigma akashiwo: Growth and uptake kinetics in laboratory cultures. Harmful Algae 6: 260-270.
- Berg GM, Glibert PM, Lomas MW, et al. (1997) Organic nitrogen uptake and growth by the chrysophyte Aureococcus anophagefferens during a brown tide event. Marine Biology 129: 377-387.
- Fan C, Glibert P, Burkholder J (2003) Characterization of the affinity for nitrogen, uptake kinetics, and environmental relationships for Prorocentrum minimum in natural blooms and laboratory cultures. Harmful Algae 2: 283-299.
- Howard MDA, Cochlan WP, Ladizinsky N, et al. (2007) Nitrogenous preference of toxigenic Pseudo-Nitzschia australis (Bacillariophyceae) from field and laboratory experiments. Harmful Algae 6: 206-217.
- Balangoda A (2016) Artificial destratification effects on nitrogen and phosphorus dynamics in a eutrophic impoundment in the Northern Great Plains. Environ Monit Assess 188: 469.
- APHA (2005) Standard methods for the examination of water and wastewater. American Public Health Association, American Water Works Association, Water Environmental Federation.
- Balangoda A, Padmanabhan G, Rahman S (2015) Effects of intermittent artificial circulation in summer months on chlorophyll a concentration in a small eutrophic impoundment. American Journal of Environmental Sciences 11: 380-391.
- Jones JR, Hoyer MV (1982) Sportfish harvest predicted by summer chlorophyll a concentration in Mid -western lakes and reservoirs. Trans Am Fish Soc 9: 111-176.
- Heyman ULF, Lundgren ALF (1988) Phytoplankton biomass and production in relation to phosphorus. Hydrobiologia 170: 211-227.
- Kalchev RK, Beshkova MB, Boumbarova CS, et al. (1996) Some allometric and non-allometric relationships between chlorophyll a and abundance variables of phytoplankton. Hydrobiologia 341: 235-245.
- Dillon PJ, Rigler FH (1974) The phosphorus chlorophyll relationship in lakes. Limnology and Oceanography 19: 767-773.
- Elser JJ, Goldman CR (1991) Zooplankton effects on phytoplankton in lakes of contrasting trophic status. Limnology and Oceanography 36: 64-90.
- Mazumder A (1994) Phosphorus-chlorophyll relationships under contrasting herbivory and thermal stratification: predictions and patterns. Canadian Journal of Fisheries & Aquatic Sciences 51: 390-400.
- Xie P (1996) Experimental studies on the role of planktivorous fishes in the elimination of Microcystis bloom from Donghu Lake using enclosure method. Chinese Journal of Oceanology and Limnology 14: 193-204.
- Scheffer M (1998) Ecology of shallow lakes. Chapman & Hall, London, 357.
- Kufel L (1998) Chlorophyll-nutrients-Secchi disc relationships in the Great Masurian Lakes (north-eastern Poland). Polish Journal of Ecology 46: 327-337.
- Kufel L (1999) Dimictic versus polymictic Masurian lakes: similarities and differences in chlorophyll - nutrients- SD relationships. Hydrobiology 408: 389-394.
- McCauley E, Downing JA, Watson S (1989) Sigmoid relationships between nutrients and chlorophyll among lakes. Canadian Journal of Fisheries & Aquatic Sciences 46: 1171-1175.
- Watson S, McCauley E, Downing JA (1992) Sigmoid relationships between phosphorous, algal biomass, and algal community structure. Canadian Journal of Fisheries and Aquatic Science 49: 2605-2610.
- Berman T, Chava S, Kaplan B, et al. (1991) Dissolved organic substrates as phosphorus and nitrogen sources for axenic batch cultures of freshwater green algae. Phycologia 30: 339-345.
- Glibert PM, Heil CA, Hollander D, et al. (2004) Evidence for dissolved organic nitrogen and phosphorus uptake during a cyanobacterial bloom in Florida Bay. Marine Science Faculty Publications 280: 73-83.
- Qin C, Liu HZ, Liu L, et al. (2015) Bioavailability and characterization of dissolved organic nitrogen and dissolved organic phosphorus in wastewater effluents. Sci Total Environments 511: 47-53.
Corresponding Author
Anusha Balangoda, Environmental and Conservation Sciences, North Dakota State University, Fargo, North Dakota, USA, Tel: 701-540-2227.
Copyright
© 2017 Balangoda A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.