Dehydrated Human Amnion/Chorion Membrane Allograft for Treatment of Recalcitrant Pyoderma Gangrenosum in PASH Syndrome and Systemic Lupus Erythematosus
Abstract
Pyoderma gangrenosum (PG) is a rare, sterile, inflammatory neutrophilic dermatosis that is painful with necrotizing ulcerations. It can be idiopathic or in association with systemic disease such as PG, acne, hidradenitis suppurativa (PASH) syndrome. PASH syndrome itself is rare, consists of multiple neutrophilic dermatoses, and is thought to be caused by an underlying genetic predisposition. Due to recurrence and poor healing, treatment for PG ranges from wound care to topical and oral medications to surgical procedures. Usually, a combination of most, if not all, of these therapies is necessary to resolve PG. Some patients, however, are resistant to many of the currently used treatment options and live with debilitating pain and extensive wound care regimens. Human amniotic tissue has been used to treat a variety of wounds over the last century. However, it has rarely been utilized in the setting of treatment resistant PG with only two other cases reported in the literature. The patient presented in this case developed worsening PG in the setting of PASH syndrome and systemic lupus erythematosus that was refractory to multiple treatment methods. After undergoing four allograft treatments with dehydrated human amnion/chorion membrane, the patient began to notice drastic improvements of her PG.
Keywords
Pyoderma gangrenosum, Systemic lupus erythematosus, Dehydrated human amnion/chorion membrane allograft, PASH syndrome, Poor wound healing
Background
Pyoderma gangrenosum (PG) is an uncommon, inflammatory neutrophilic dermatosis characterized by recurrent flare-ups and is sterile with painful, necrotizing ulcerations [1]. PG can be isolated or associated with infection, systemic diseases such as Systemic lupus erythematosus (SLE), inflammatory bowel disease, rheumatoid arthritis, hematologic disorders, or malignancy [2]. It can also be part of autoinflammatory syndromes such as PG, acne, and hidradenitis suppurativa (HS) (PASH) syndrome [3]. PG pathophysiology is poorly understood and treatment typically includes topical and systemic immunosuppression combined with wound care [1]. However, PG may be debilitating due to treatment failure. In this case, a patient with SLE develops treatment resistant PG associated with PASH syndrome and is successfully treated with dehydrated human amnion/chorion membrane (dHACM) allograft therapy.
Case Presentation
The patient is a 36-year-old African American female with a history of SLE treated with oral hydroxychloroquine 200 mg twice daily and oral prednisone 10 mg once daily who presented to clinic for an ulcer on her left lower extremity. She banged her left leg, which initially bruised then ulcerated. It did not heal despite topical bacitracin ointment. Physical examination of her left lower leg revealed a small ulceration with rolled edges and surrounding erythema just above the medial malleolus. It was recommended to apply wound dressings, bacitracin, and see wound care.
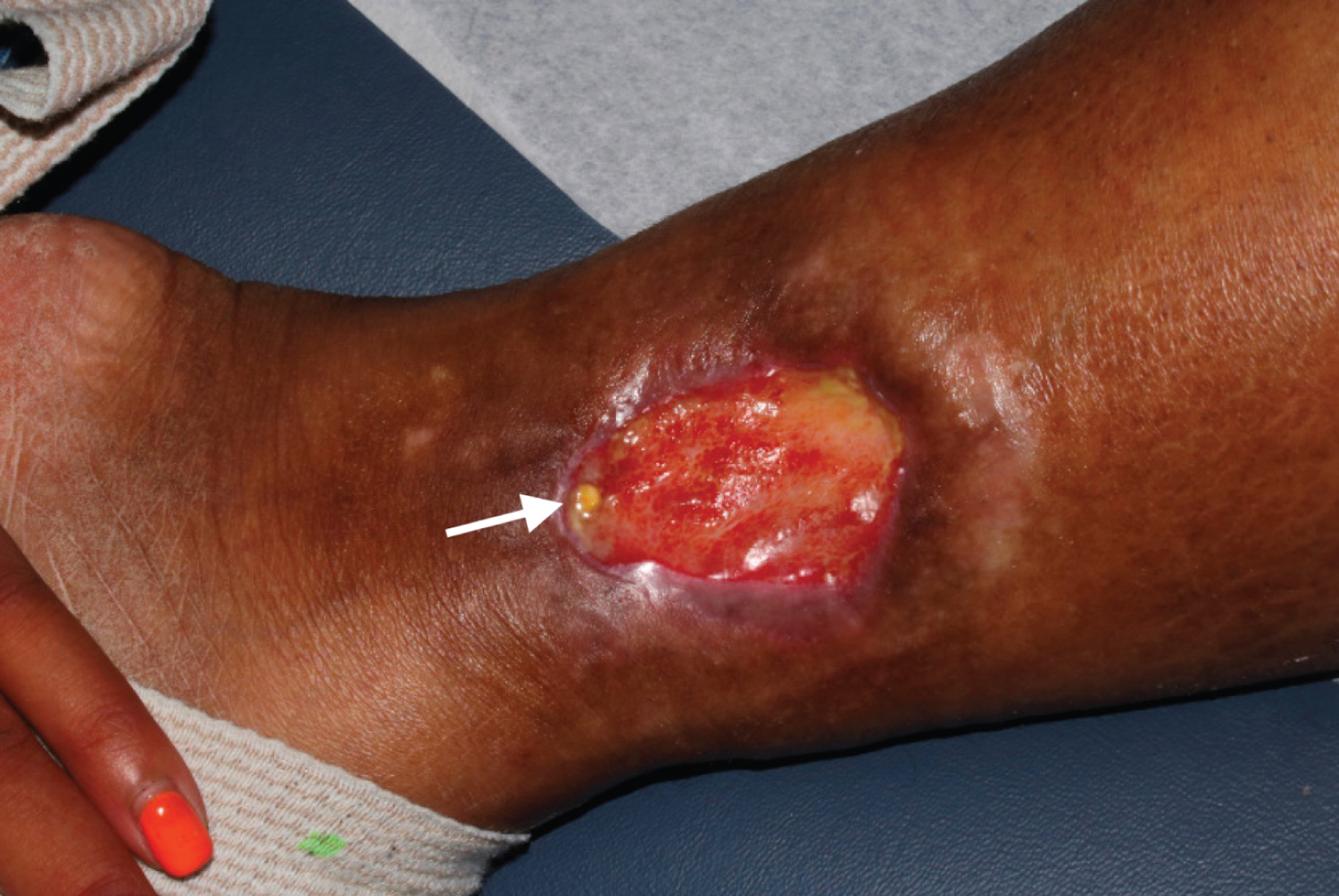
Over the next several months, her ulcer enlarged and became extremely tender. There was now a deep ulcer with an erythematous border surrounding the wound edge (Figure 1). Additionally, three small subcutaneous nodules with small sinus tracts were noticed in her left maxilla and groin. She was diagnosed PASH syndrome.
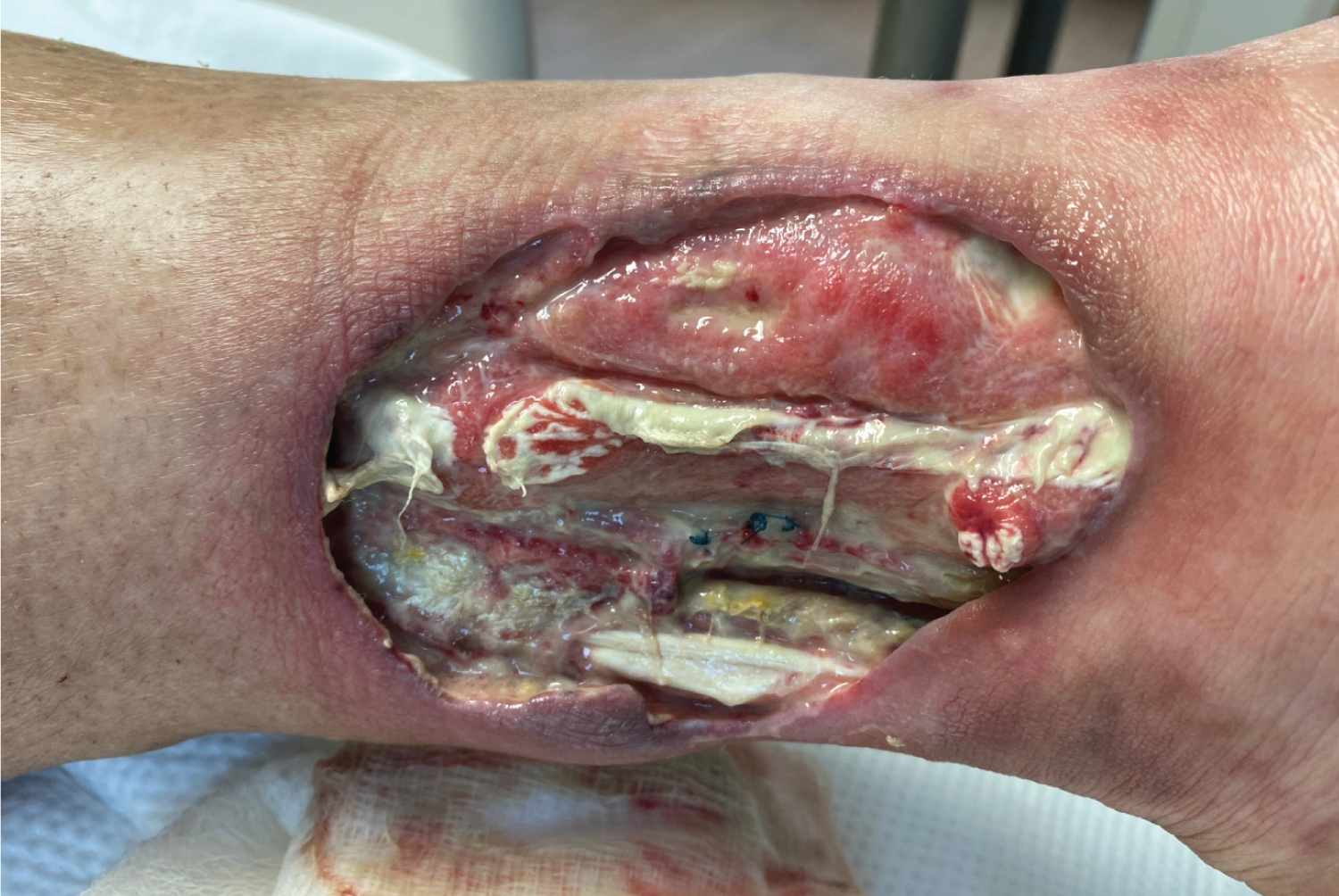
The patient tried several treatment regimens over two years including increasing her daily oral prednisone, azithromycin, clindamycin, doxycycline, dapsone, adalimumab, cyclosporine, timolol 0.25% drops, clobetasol 0.05% ointment, and frequent dressing changes as well as hyperbaric therapy through wound care. Adalimumab improved her HS, but her PG worsened. The care team decided to debride the wound and inject intralesional triamcinolone. Post-operation follow-up examination revealed a large, deep ulceration in the left medial lower leg that was now extending towards the posterior calf with undermining borders and exposure of tendon. There were sparse, focal areas of granulation tissue with overlying purulent drainage and diffuse lower extremity edema (Figure 2).
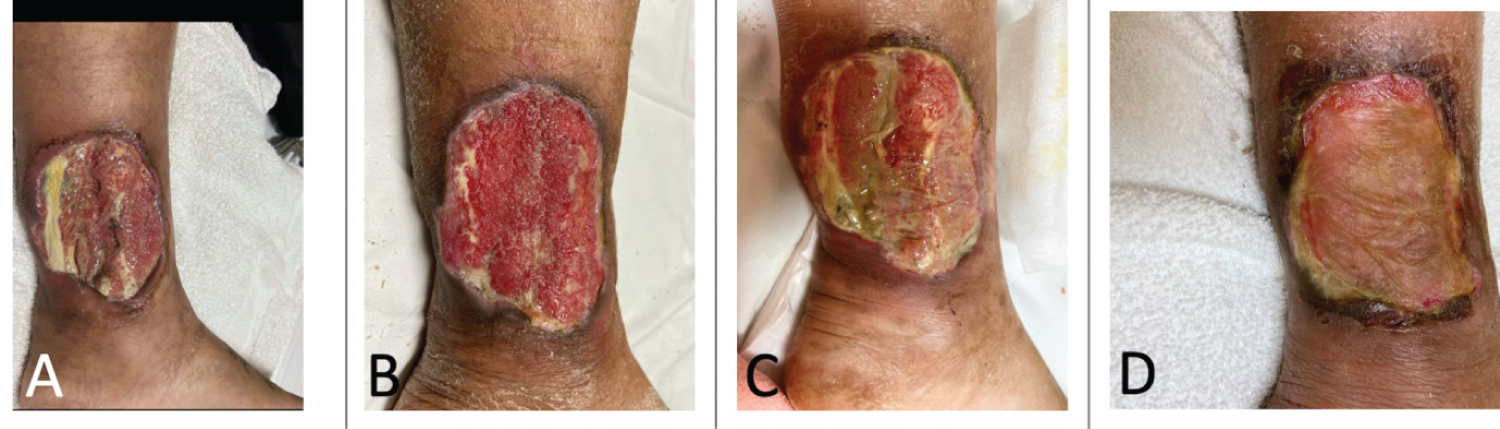
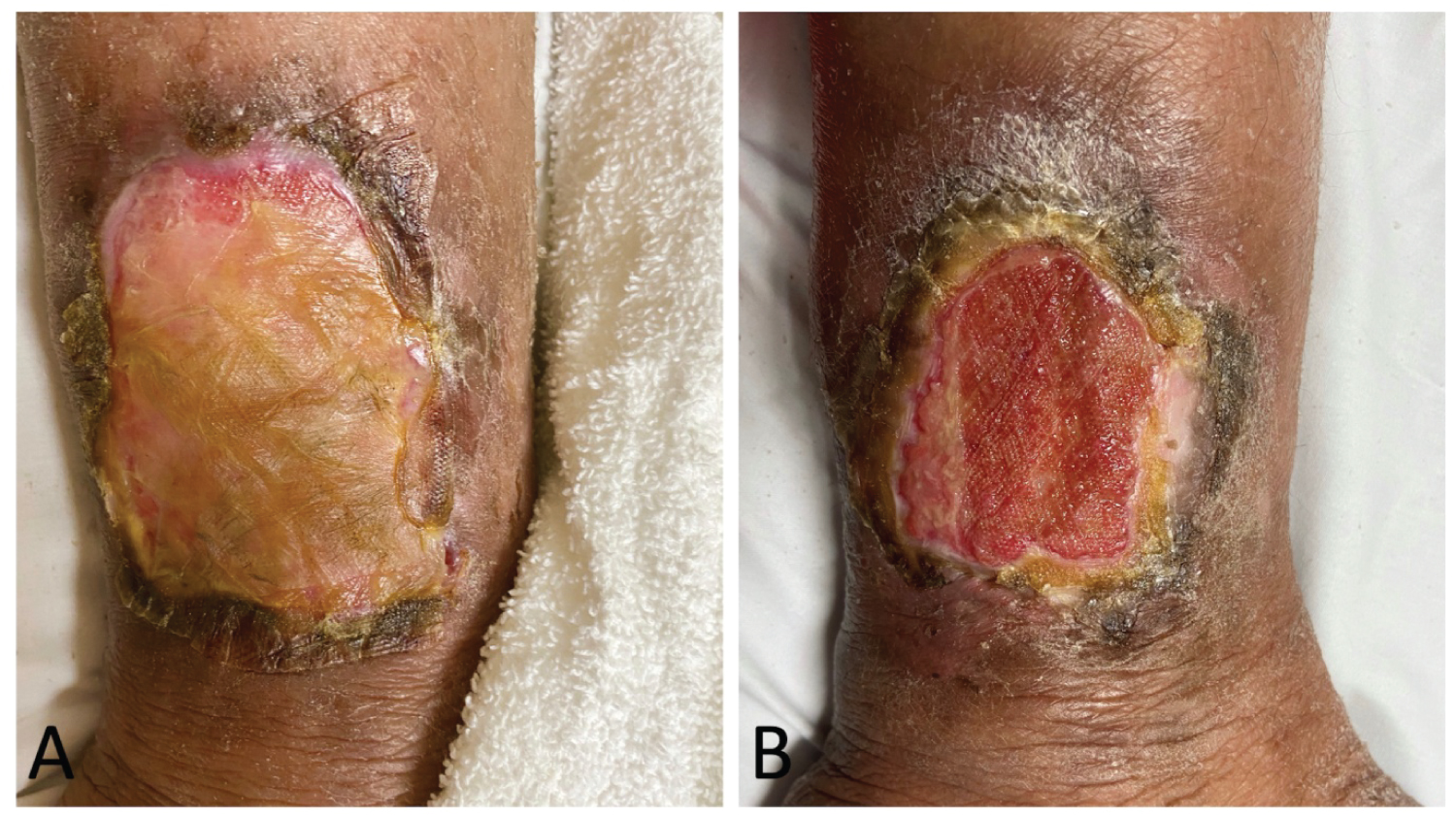
The PG remained unresponsive and continued to worsen, and a decision was made to use dHACM allograft therapy. After the first treatment, the patient had notable improvement in pain and functionality. She received three additional rounds of dHACM over a total of four months (Figure 3). Her chronic ulcer that measured 10 × 9 × 0.4 cm just prior to treatment reduced to 8 × 6 × 0.1 cm after dHACM therapy and continues to improve in conjunction with her hydroxychloroquine, maintenance dose oral prednisone, topical medications, and wound care (Figure 4).
Conclusion
PG as an entity of PASH syndrome is uncommon. PASH syndrome is a distinct autoinflammatory disease comprised of multiple neutrophilic dermatoses that likely has underlying genetic predisposition [4,5]. It has recently been recognized in the literature; thus, there are no evidence-based treatment guidelines to date. A recent case report describes PASH syndrome successfully treated with adalimumab, antibiotics, weight loss, and intensive wound care and dressing changes [6].
PG, regardless if isolated or part of other systemic syndromes, can be difficult to treat given the recalcitrant nature of the disease; hence, many treatment combinations are utilized. Topical therapies such as corticosteroids and calcineurin inhibitors are often prescribed [1]. Surgical management can be complicated by pathergy, which is described as skin trauma that initiates a new PG lesion or exacerbates a preexisting one. Therefore, the utility and effectiveness of surgical therapy is controversial [7].
Systemic immunosuppressive agents may be prescribed such as cyclosporine, azathioprine, dapsone, methotrexate, tacrolimus, corticosteroids, TNF-alpha inhibitors, mycophenolate mofetil, intravenous immunoglobulins, cyclophosphamide, rituximab, alefacept, anakinra, canakinumab, and ustekinumab. Antibiotics are occasionally used in conjunction with systemic agents [1]. Additionally, negative pressure wound therapy has been shown to facilitate nonhealing wounds in the lower extremities [8].
Treatment of PG with dHACM is rarely reported in the literature and may be a viable option for those who have failed prior treatments. dHACM has been used in a variety of clinical settings including wound healing and can assist in decreasing inflammation and scar tissue development, reducing pain, increasing angiogenesis, and providing a cellular proliferation and migration matrix [8]. dHACM is thought to be superior to live skin substitutes for several reasons. dHACM acts as a "stem cell magnet" to stimulate hematopoietic or mesenchymal cells that are brought to injured sites through soluble mediators in the wound repair process. Furthermore, an allograft that consists of chorion and amnion has been shown to have increased growth factors and cytokines that may assist in more complete and rapid healing. This therapy may also prove more cost effective than living cellular skin substitutes because it is easier to form the graft to the wound, resulting in less wasted graft material. Also, fewer grafts are required to achieve complete healing [9].
While dHACM has been reported in the treatment of diabetic and venous ulcers, it has rarely been reported in the treatment of PG. In 1978, Gruss reported the first successful treatment of PG using amniotic membrane with a reduction in wound size of 66% [10]. Then, in 2015, Snyder, et al. reported the first contemporary use of dHACM in PG with similar results [8]. To our knowledge, these are the only two cases that have previously reported treatment of PG with dHACM. Thus, this case of a patient with SLE and PASH syndrome who is treated with dHACM after several failed attempts to treat PG adds to the very limited literature. After several rounds of allograft therapy, the patient's PG is successfully resolving and no longer causes her pain.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Acknowledgements
None.
Funding
The author(s) receive no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Quist SR, Kraas L (2017) Treatment options for pyoderma gangrenosum. JDDG: Journal der Deutschen Dermatologischen Gesellschaft 15: 34-40.
- Ruocco E, Sangiuliano S, Gravina AG, et al. (2009) Pyoderma gangrenosum: An updated review. Journal of the European Academy of Dermatology and Venereology 23: 1008-1017.
- Cugno M, Borghi A, Marzano AV, et al. (2017) PAPA, PASH and PAPASH syndromes: Pathophysiology, presentation and treatment. American Journal of Clinical Dermatology 18: 555-562.
- Braun-Falco M, Kovnerystyy O, Lohse P, et al. (2012) Pyoderma gangrenosum, acne, and suppurative hidradenitis (PASH)-a new autoinflammatory syndrome distinct from PAPA syndrome. Journal of the American Academy of Dermatology 66: 409-415.
- Niv D, Ramirez JA, Fivenson DP (2017) Pyoderma gangrenosum, acne, and hidradenitis suppurativa (pash) syndrome with recurrent vasculitis. JAAD Case Reports 3: 70-73.
- Zagaria O, Ruggiero A, Fabbrocini G, et al. (2020) Wound care, adalimumab, and multidisciplinary approach in a patient affected by PASH syndrome. International Wound Journal 17: 1528-1531.
- Haag CK, Bacik L, Latour E, et al. (2020) Perioperative management of pyoderma gangrenosum. Journal of the American Academy of Dermatology 83: 369-374.
- Snyder RJ, Ead J, Glick B, et al. (2015) Dehydrated human amnion/chorion membrane as adjunctive therapy in the multidisciplinary treatment of pyoderma gangrenosum: A case report. Ostomy Wound Manage 61: 40-49.
- Zelen CM, Gould L, Serena TE, et al. (2015) A prospective, randomised, controlled, multi-centre comparative effectiveness study of healing using dehydrated human amnion/chorion membrane allograft, bioengineered skin substitute or standard of care for treatment of chronic lower extremity diabetic ulcers. International Wound Journal 12: 724-732.
- Gruss JS, Jirsch DW (1978) Human amniotic membrane: A versatile wound dressing. Canadian Medical Association Journal 118: 1237-1246.
Corresponding Author
Amanda S. Weissman, BS, Medical College of Georgia, Augusta University, Augusta, GA, USA, Tel: 954-729-0513.
Copyright
© 2021 Weissman AS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.