Experimental Study of Bulk Water Removal in Two Phase Flow
Abstract
The improvement of oil-water separation efficiency has become increasingly important in the oil and gas industry, as well as for environmental protection. This study uses both experimental and numerical methods to investigate the effects of flow rate (v), oil volume fraction (vof), and temperature (T) on oil-water separation efficiency. Design Expert software is used to analyzing data of the experiments. Experimental results show that the oil volume fraction and temperature are significant factors for separation efficiency. Also the interaction effects between flow rate and oil volume fraction, and that between flow rate and temperature are significant for oil-water separation efficiency. The oil concentration in separated water decreases significantly as the oil volume fraction increases.
Keywords
Oil-water separation, Multiphase flow, Design of experiment
Introduction
Oil/water separation is important due to increasing industrial oily-waste water in the oil and gas industry [1, 2]. The traditional way to separate oil-water in the industry is by using a gravity-based separator [3,4]. Oil and water separate in the separator because of the difference in specific gravity; oil will move to the surface of the water, while water will move to the bottom of the separator. Keller [5] devised a method to study multiphase flow with oil density between 11 to 70 API gravity. In the study, multiphase flow was injected into the separator, passing through a filter media system designed to collect oil droplets. Lars Schlieper, et al. [6] conducted an experiment in 2004 to investigate the separation behavior of a horizontal gravity separator with three different inner components. This study mainly focused on how inflow to the plate, the plate material, and the distance between the plates influenced the separation length of the separator. They proved that separators with inner components, like plates, can significantly reduce separator length compared to other separators. In 2009 Fitnawan, et al. [7] introduced a new design in gravity separators: the inclined gravity down hole oil-water separator. They found that using this new structure, they were able to increase separation efficiency up to 82%. Krebs, et al. [8] used a kinetic analysis to estimate the relationship between coalescence time and mean droplet diameter in the oil and water multiphase fluid system. They stated that the growth of separated oil and mean droplet diameter followed as a function of time ex-situ to quantify the kinetics of coalescence. In the oil-water two-phase RSM model Liu, et al. [9] tested the velocity, pressure, and oil concentration distribution of the cyclone separator. The results of this simulation provided the basis for improvements and optimizations to the design of the cyclone separator structure.
Separation efficiency relies on the design of the separator and the operating conditions. This research examines the relationship between flow rate, oil volume fraction, temperature, and oil concentrations. The objective of this paper is to determine three operating factors, which is flow rate, oil volume fraction and temperature, and their effects on oil-water separation efficiency in a gravity separator.
Experimental Apparatus and Design
An American Petroleum Institute (API) separator was designed using Stokes' Law [10]. The API separator is designed based on the specific gravity difference between the oil and water, which is designed standards published by the American Petroleum Institute.Stokes' Law (Eq.1) is used to calculate the rising velocity of oil droplet from the bottom of a separator to the surface of the water.
where, is oil droplet rissing velocity in m⁄s, is oil droplet horizontal velocity in m⁄s, g is gravitational acceleration in m⁄s^2 ,ρ0 is density the of oil in kg/m^3 , ρw is the density of water in (kg⁄m^3 ), μ is dynamic viscosity in N s/m2 , R is oil droplet radius in m.
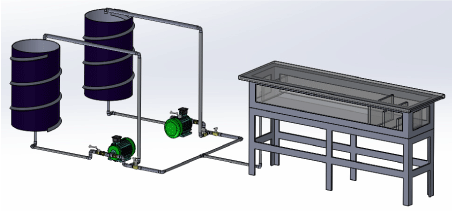
Figure 1 presents a 3D Solid Works image of the experimental setup. The properties of oil and water in this study are given in (Table 1). Both oil and water were transferred from the storage tanks with a pump to the test section made of 1-inch PVC pipe. Oil and water entered the test section from two pipes via a T-junction. Two pressure sensors and flow meters with a maximum capacity of 10 GPM were located in each of the flow lines (water and oil). After the pumps, each fluid had a bypass pipe to control the flow rate in the main line. The multiphase fluid flowed into the separator tank, where the oil and water were separated.
The traditional one-factor-at-a-time (OFAT) method has very limited ability when there are many factors for an experiment. Using OFAT method to examine the interactions between flow rate, oil volume fraction and temperature requires a large number of experiments, and it is also hard to find interactions between the factors [11,12]. Therefore, the Design of Experiment (DOE) method has been used. DOE is a methodology for systematically and statistically design experiments and analyzes data. The analysis of variance (ANOVA) method is used to analyze the data [13]. DOE is more efficient and capable and is able to reduce the number of experiments significantly without losing the desired data points.
The main purpose of this paper is to study the interaction between variables, therefore, a two-level, three-factor, face-centered composite design (23) from the response surface methodology (RSM) was used to investigate the effects of three factors: flow rate (v), oil volume fraction (vof), and temperature (T) on the response of the oil concentration. The RSM method is the most popular mythology in DOE to design three variables with maximum information and minimum experimental tests. The factors and levels are shown in Table 2. A specific run sheet was generated by the Design Expert Software. The oil concentration was calculated by taking nine 25 ml samples for each run, then an analytical balance was used to weigh the samples, Wtotal. A centrifuge separator was used to separate the oil and water samples completely. The separation operated at 8500 rpm, for 25 min for each sample. After the second separation process, water was removed from the bottom of the samples with a syringe pump. Then the analytical balance was used to measure the weight of oil, Woil.
In most of the real cases, the oil volume fraction is less than 0.5 in the oil-water multiphase flow. The minimum oil volume fraction 0.2 was designed according to the measurement range of flow meter, which is from 0.1 to 10 GPM. Base on the temperature limitation of glue which is used to connect the pipeline of this experimental setup, temperature range from 20℃ to 32℃ was selected in this study. In this study, the oil-water separation efficiency is represented by oil concentration.
According to Kline and McClintock [14], in general, if n measurements vn are being made, each with a measurement tolerance of wn, and a function R is calculated using the measured values, then the uncertainty or tolerance in the calculation can be determined as:
Based on Eq. (4), for the oil concentration uncertainties of this experiment, Eq. (3) only has one variable, therefore, wR is determined by the accuracy of the analytical balance, which around 0.0001.
Results and Discussion
After completing the experiments based on the run sheet (a total of 20 runs), an ANOVA result was generated by Design Expert. The ANOVA table is shown in Table 3. Sum of square (SS) means variability of all the experimental data. The smaller value of SS likely indicates the better the model's estimation will be. df represents the degree of freedom. Mean square is a sum of square divided by its df. A p-value less than 0.0500 indicates the factor is significant. Based on this result, some significant factors are as follows: flow rate, oil volume fraction, the interaction effects between flow rate and oil volume fraction, the interaction effects between flow rate and temperature, and the interaction effects between oil volume fraction and temperature.
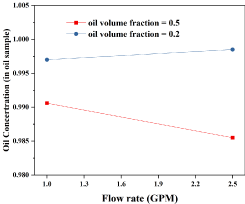
According to Figure 2, when the oil volume fraction = 0.5, the oil concentration decreases as the flow rate increases. However, when the oil fraction = 0.2, oil concentration increases slightly with the increases of the flow rate. With the increase of oil volume fraction, the oil concentration decreases.
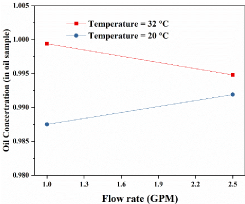
Figure 3 indicates that flow rate and temperature have complex effects on oil concentration. When temperature T= 32℃, oil concentration decreases significantly with the increase of flow rate. However, when the temperature is 20℃, the flow rate has positive impacts on oil concentration. With the increase of temperature, the oil concentration decreases.
As shown in Figure 2 and Figure 3, under lower oil volume fraction and lower temperature, separation efficiency improved with the increase of flow rate. The possible reasons for this result were explained by Carlos, et al. [15] in 2001. One reason is a higher flow rate means shorter residence time but higher acceleration forces. However, lower flow rates mean longer residence times but lower acceleration forces. In addition, Jiang, et al. [16] and Liu, et al. [17] found that when the flow rate is lower than 20 GPM, the separation efficiency increases with the flow rate. As they explained, another reason is that large flow rate breaks oil droplets into smaller ones, and this increases the emulsification degree of the multiphase fluid. This discourages the separation. On the other hand, a higher flow rate improves the motion of the liquid inside a separator, which is beneficial for oil-water separation.
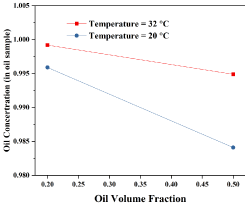
Figure 4 shows that oil volume fraction has negative effects on oil concentration. With the change of the oil volume fraction from 0.1 to 0.5, oil concentration decreases sharply. Increasing temperature also results in the increase of oil concentration. Reyes, et al. [18] proved a similar oil volume fraction influence on the separation efficiency trend by using both the simulation and experimental methods in 2006.
Both Figure 3 and Figure 4 show that higher temperature leads to better separation efficiency. The same result is found by Kokal and Al-Ghamdi in 2005 [19]. One of the factors that influence the oil-water separation efficiency is the stability of the emulsion. Based on their study, the oil-water emulsion stability decreases with the increase of temperature. Therefore, higher temperature helps better separation efficiency.
Conclusions
An API designed separator was used to investigate the effects of flow rate (v), oil volume fraction (vof), and temperature (T) on oil concentration. By using the statistical design of experiments, a two-level, three-factor, face-centered composite method was used to investigate the effect on separation efficiency. Based on the ANOVA results, the oil volume fraction (vof) is one of the most important factors on separation efficiency. Oil concentration decreases as the oil volume fraction increases. The interaction effects of flow rate and temperature on oil concentration are complex. Under a higher temperature, the oil concentration decreases with flow rate. However, for a lower temperature, the oil concentration increases with flow rate. This result is consistent with other previous studies.
Acknowledgments
The authors wish to thank the Dr. Zongming Wang for his support on building experimental setup. The authors are also thankful for the financial support from the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Research & Development Corporation (RDC) support for building the flow loop.
References
- Xue Z, Wang S, Lin L, et al. (2011) A novel superhydrophilic and underwater superoleophobic hydrogel-coated mesh for oil/water separation. Advanced Materials 23: 4270-4273.
- Al-Yaari MA, Abu-Sharkh BF (2011) CFD prediction of stratified oil-water flow in a horizontal pipe. Asian Trans Eng 1: 68-75.
- Hinze JO (1955) Fundamentals of the hydrodynamic mechanism of splitting in dispersion processes. AIChE Journal 1: 289-295.
- Crowe CT, Schwarzkopf JD, Sommerfeld M, et al. (2011) Multiphase flows with droplets and particles. CRC press.
- Keller Jr. HF (1975) oil-Water Separation Process. U.S. Patent 3867285.
- Schlieper L, Chatterjee M, Henschke M, et al. (2004) Liquid-liquid phase separation in gravity settler with inclined plates. AIChE Journal 50: 802-811.
- Fitnawan E, Rivera R, Golan M (2009) Inclined Gravity Down hole Oil-Water Separator: Using Laboratory Experimental Results for Predicting the Impact of Its Application in High Rate Production Wells. In Asia Pacific Oil and Gas Conference & Exhibition.
- Krebs T, Schroen CGPH, Boom RM (2021) Separation kinetics of an oil-in-water emulsion under enhanced gravity. Chemical Engineering Science 71: 118-125.
- Liu HF, Xu JY, Wu YX (2010) Numerical study on oil and water two-phase flow in a cylindrical cyclone. Journal of Hydrodynamics 22: 832-837.
- Huang HH, Bouffard S, Su Y Oil-Water Separation System for Industrial Wastewater.
- Fisher RA (1935) The design of experiments. New York: Hafner Publishing Company.
- Myers RH, Montgomery DC (2016) Process and product optimization using designed experiments: Response surface methodology.
- Lye LM (2002) Design of Experiments in Civil Engineering: Are we still in the 1920s. In Proceedings of the 30th Annual Conference of the Canadian Society for Civil Engineering Montreal, Quebec.
- Kline SJ, McClintock FA (1953) Describing uncertainties in single-sample experiments. Mechanical Engineering 75: 3-8.
- Gomez C, Caldentey J, Wang S, et al. (2001) Oil-water Separation in Liquid-liquid Hydrocyclones (LLHC)-experiment and Modeling. SPE Annual Technical Conference and Exhibition. Society of Petroleum Engineers.
- Minghu J, Zhao L (2007) Pressure and separation performance of oil/water hydrocyclones. Production and Operations Symposium. Society of Petroleum Engineers
- Li HF, Xu JY, Zhang J, et al. (2012) Oil/water separation in a liquid-liquid cylindrical cyclone. Journal of Hydrodynamics Ser. B 24: 116-123.
- Reyes MA, Pacheco JE, Marín JC, et al. (2006) Numerical simulation and experiments of the multiphase flow in a liquid-liquid cylindrical cyclone separator. Proceedings of FEDSM 2006 ASME Joint US-European Fluids Engineering Summer Meeting.
- Kokal SL, Al Ghamdi A (2006) Oil/water separation experience from a large oil field. SPE Production & Operations 21: 365-371.
Corresponding Author
Weiwei E, Memorial University of Newfoundland, 230 Elizabeth Ave, St. John's, NL A1C 5S7, Canada.
Copyright
© 2021 Weiwei E. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.