Diffuse Leptomeningeal Glioneuronal Tumor: Case Report and Review of Literature
Abstract
In this study, we describe a rare case of disseminated CNS malignancy of childhood. A seven-year-old boy with headache, vomiting and gait disturbances was diagnosed with non-obstructive hydrocephalus and papilledema. Initial MRI revealed no significant findings, however subsequent studies revealed bilateral extradural and leptomeningeal enhancement. Biopsy was eventually obtained with pathology report confirming leptomeningeal oligodendrogliomatosis. We discuss the recent description in literature of this rarely reported diagnosis. This entity primarily affects pediatric patients. It has a unique clincopathological identity, which can be easily mistaken for an inflammatory process. The diagnosis of this tumor can be complicated due to the rather nonspecific clinical presentation and findings on imaging.
Keywords
Oligodendrogliomatosis, Leptomeningeal spread, CNS malignancy, Glioneural tumor
Introduction
Disseminated CNS malignancies of childhood are rare. Until recently only a few cases were described in the literature. In recent years, however, reports on pediatric cases with distinct pathological and molecular features led to the creation of a new tumor entity called diffuse leptomeningial glioneuronal tumor [1-5].
The clinical diagnosis of this tumor is sometimes complicated due to the rather nonspecific imaging findings [6]. On MRI these lesions usually present as diffuse thickening and enhancement of the dura of both the brain and spinal cord [6]. Often times this lesion is confused with an inflammatory process such as neruosarcoidsosis [7]. The clinical presentation is also rather nonspecific and can include focal neurological deficits, headaches due to increased intracranial pressure and changes in mental status [1-3,7].
In this report we present the case of 7-year-old boy with diffuse leptomeningeal oligodendrogliomatosis. As with many clinical scenarios of this disease, the initial clinical presentation is nonspecific. The progress of disease is slow with imaging findings developing sometime after the initial clinical presentation. We also report the full pathology and molecular work up of the lesion and make comparisons to the literature describing other clinical presentations of this tumor.
Case Presentation
A 7-year-old boy presented with a five-day history of headaches, vomiting, and gait disturbance. Examination revealed bilateral papilledema and cranial CT showed dilated ventricular system indicative of non-obstructive hydrocephalus. MRI confirmed the CT findings and did not display any brain parenchymal changes or leptomeningeal enhancement. It should be noted that six months prior to admission, a non-contrast enhanced CT scan, performed at another institution for mild head trauma, already showed ventricular dilatation. A Ventriculo-Peritoneal shunt was inserted, and the boy was discharged from hospital five days later. Cytological examination of the CSF was negative. Examination 3 months later, showed that the papilledema had resolved.
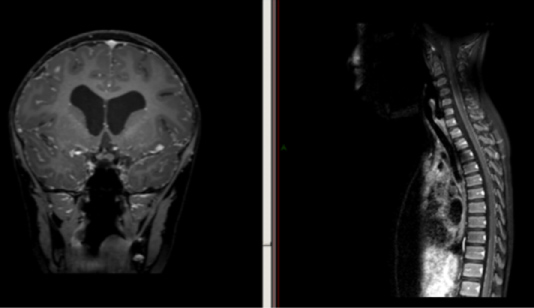
Four months following surgery the boy presented again with headaches and left leg weakness. A MRI revealed small extra axial collections and diffuse meningeal enhancement. He was subsequently discharged with instructions for follow up. Several months later, the boy returned with headaches and repeated falls. MRI of the head and spine showed ventriculomegaly and leptomeningeal enhancement of the cranial leptomeninges with a nodular lesion in the left sylvian fissure and a linear enhancement of the spinal leptomeninges (Figure 1). Since repeated lumbar punctures and shunt taps resulted in negative CSF cytology. A biopsy of the right frontal meninges was performed. The pathological diagnosis was disseminated oligodendroglial - like leptomeningeal tumor.
The boy was treated with temozolomide and greatly improved clinically, although radiographic improvement was minimal. However, nine months after treatment beginning his headaches and vomiting recurred and imaging showed increased enhancement, in particular the cerebellar folia. He was then entered on a phase I clinical trial of a MEK inhibitor.
Pathological Findings
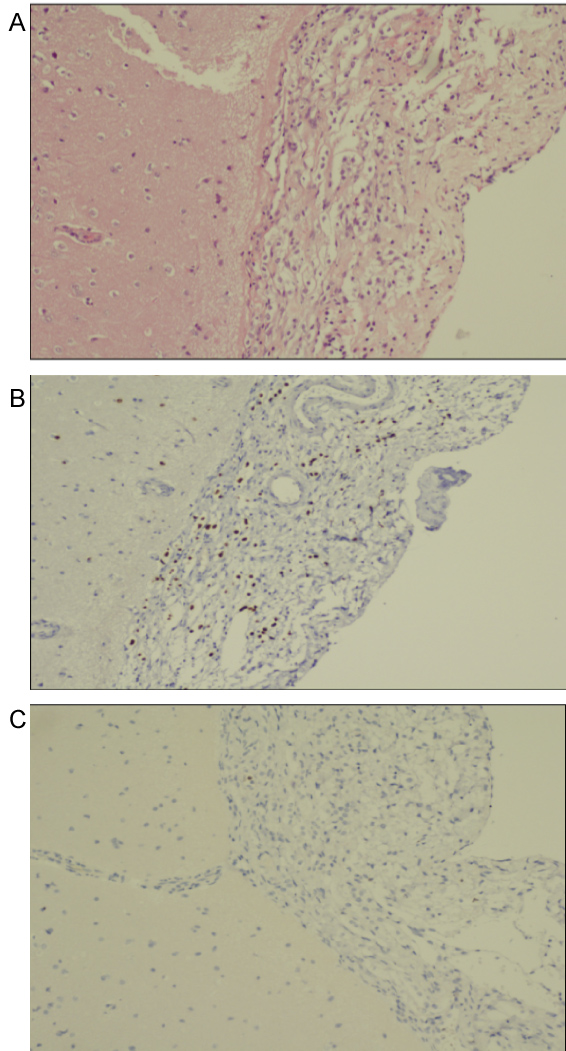
Histological examination revealed infiltration of the leptomeninges by scattered cells with round nuclei and clear cytoplasm. The cells displayed no significant atypia and no mitotic figures were identified. The neoplastic cells were immune positive, for OLIG2 and, S100 protein and also for GFAP and MAP2 (Figure 2). They did not stain for synaptophysin, IDH1-R132H and p53, A diagnosis of disseminated oligodendroglial - like leptomeningeal tumor was made. Molecular analysis demonstrated a BRAF- KIAA1549 fusion, confirming the pathological diagnosis.
Discussion
We present a case of a 7-year-old boy with diffuse leptomeningeal glioneuronal tumor. As with many cases of this disease, the presentation is nonspecific, and the clinical evolution is slow. The initial physical manifestations of this disease are varied, including focal neurological signs of increased intracranial pressure, convulsions and altered mental status. This entity may mimic non-tumoral diseases like neurosarcoidosis or infectious agents that can cause chronic meningitis [7].
The entity was first described by Beck and Russel in 1942 in which they described four cases of "Oligodendrogliomatosis of cerebrospinal pathaway" [8]. A series of 36 cases found high proportion of pediatric patients with a male predominance in 66% [2]. Similar to the patient that we presented, cytology form the CSF in this series was negative in 16 patients, indicating that definitive diagnosis could only be obtained by biopsy [2]. However, non-diagnostic brain, pia and arachnoid biopsies are a common phenomena and sometimes several attempts are necessary to arrive at the final diagnosis. As mentioned the CSF samples are typically negative for malignant cells and have a high protein concentration [7]. Lesions of this disease are mainly located in the subarachnoid space; it is unclear whether its origin is in the leptomeningeal compartment or leptomeningeal extension of a subtle intraparenchymal disease [3].
Histologically this lesion shows a classic oligodendroglial pattern: Round cells with surrounding halos [2]. In a minority of cases, astrocytic features are also seen [2]. Immunohistochemistry studies show positive staining for OLIG2 in 100% of cases with staining for other molecules such as S100, GFAP, synaptophysin, and chromogranin A also present [2,9]. On the molecular level previous studies have shown both 1p19q codeletion at rate of about 20%, however there are no phenotypic differences based on deletion status of this gene [2,10,11]. In the case presented in this study, tissue sample stained positive for the above-mentioned markers while at the same time showing 1p19q codeletion. It is also interesting to note that the presence of BRAF-KIAA1549 fusion gene was also detected using PCR. The presence of this gene has been shown to have good prognostic outcome while at the same time serving as a possible therapeutic target , however future studies are needed for validation [12,13].
The radiological features of this disease show diffuse leptomeningeal involvement with enhancement throughout the brain and spine on MRI studies [2]. The presence of concomitant hydrocephalus is not uncommon. In the initial stages of disease, the enhancement of the spine is thin, and it looks as the nerves roots are coated with paint [2]. As the disease progresses the enhancement in both the brain and spinal cords shifts from the subarachnoid space to the intra-axial compartment and the lesions become more nodular in nature [6,14]. There are reports of cystic lesions in the posterior fossa which are hyperintense on T2 weighted imaging [2]. The imaging findings of this lesion are rather nonspecific, and it is difficult to make the diagnosis alone based on radiological findings.
In this study we have presented the case of 7-year-old male patient with diffuse leptomeningeal oligodenrogliomatosis. The initial presenting symptoms were rather nonspecific as were the radiological findings. As we have described this lesion is a complicated entity, which can be mistaken for infectious processes. However, we emphasize to clinicians that leptomeningeal oligodendrogliomatosis of childhood must be kept in the differential diagnosis with patients presenting with nonspecific neurological findings in combination with diffuse subarachnoid enhancement on brain and spine MRI.
References
- Mathews MS, Pare LS, Kuo JV, et al. (2009) Primary leptomeningeal oligodendrogliomatosis. J Neurooncol 94: 275-278.
- Rodriguez FJ, Perry A, Rosenblum MK, et al. (2012) Disseminated oligodendroglial-like leptomeningeal tumor of childhood: A distinctive clinicopathologic entity. Acta Neuropathol 124: 627-641.
- Preuss M, Christiansen H, Merkenschlager A, et al. (2015) Disseminated oligodendroglial cell-like leptomeningeal tumors: Preliminary diagnostic and therapeutic results for a novel tumor entity. J Neurooncol 124: 65-74.
- Chellathurai A, Vaidya JS, Kathirvelu G, et al. (2016) Primary diffuse leptomeningeal oligodendrogliomatosis: A case report and literature review. Indian J Radiol Imaging 26: 337-341.
- Louis DN, Perry A, Reifenberger G, et al. (2016) The 2016 world health organization classification of tumors of the central nervous system: A summary. Acta Neuropathol 131: 803-820.
- Kessler BA, Bookhout C, Jaikumar S, et al. (2015) Disseminated oligodendroglial-like leptomeningeal tumor with anaplastic progression and presumed extraneural disease: Case report. Clin Imaging 39: 300-304.
- Hunderfund ANL, Zabad RK, Aksamit AJ, et al. (2013) Diffuse anaplastic leptomeningeal oligodendrogliomatosis mimicking neurosarcoidosis. Neurol Clin Pract 3: 261-265.
- Beck DJK, Russell DS (1942) Oligodendrogliomatosis of the Cerebrospinal Pathway. Brain 65: 352-372.
- Dyson K, Rivera-Zengotita M, Kresak J, et al. (2016) FGFR1 N546K and H3F3A K27M mutations in a diffuse leptomeningeal tumour with glial and neuronal markers. Histopathology 69: 704-707.
- Rodriguez FJ, Schniederjan MJ, Nicolaides T, et al. (2015) High rate of concurrent BRAF-KIAA1549 gene fusion and 1p deletion in disseminated oligodendroglioma-like leptomeningeal neoplasms (DOLN). Acta Neuropathol 129: 609-610.
- Schniederjan Matthew J, Alghamdi Sarah, Castellano-Sanchez Amilcar, et al. (2013) Diffuse Leptomeningeal Neuroepithelial Tumor: 9 Pediatric Cases with Chromosome 1p/19q Deletion Status and IDH1 (R132H) Immunohistochemistry. Am J Surg Pathol 37: 763-771.
- Hawkins C, Walker E, Mohamed N, et al. (2011) BRAF-KIAA1549 fusion predicts better clinical outcome in pediatric low-grade astrocytoma. Clin Cancer Res 17: 4790-4798.
- Kumar A, Pathak P, Purkait S, et al. (2015) Oncogenic KIAA1549-BRAF fusion with activation of the MAPK/ERK pathway in pediatric oligodendrogliomas. Cancer Genet 208: 91-95.
- Armao DM, Stone J, Castillo M, et al. (2000) Diffuse leptomeningeal oligodendrogliomatosis: Radiologic/pathologic correlation. Am J Neuroradiol 21: 1122-1126.
Corresponding Author
Neelan J Marianayagam, MD, PhD, Department of Neurosurgery, Shaare Zedek Medical Center, Jerusalem, Israel.
Copyright
© 2020 Marianayagam NJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.