Myeloid Sarcoma in the Ear Followed by Labyrinthine Hemorrhage: A Rare Presentation of Late Relapse in Acute Myeloid Leukemia
Abstract
Myeloid sarcoma (MS) is an immature myeloid cells tumor that might occur in acute myeloid leukemia (AML) even without bone marrow involvement. We present a rare case of an 18-year-old teenager who developed MS in the external auditory canal (EAC), and subsequent inner ear impairment, as the first manifestation of recurrence of promyelocytic AML. High suspicion and early identification are paramount in terms of overall prognosis of the underlying condition.
Keywords
Myeloid sarcoma, Acute myeloid leukemia, External auditory canal, Labyrinthine hemorrhage
Introduction
A wide spectrum of diseases can affect the external auditory canal (EAC), including infections, congenital malformations, cholesteatoma, and benign or malignant neoplasms. Among neoplasms of the EAC, the occurrence of metastases is extremely rare and scarcely described [1,2]. In most cases, the diagnosis of EAC diseases is based on clinical features. However, both benign and malignant diseases might present with a soft tissue mass in the canal, which are often clinically indistinguishable. Complementary investigation is mandatory in patients who do not improve with the initial management and in those with a previous history of oncological disease or immunosuppression [1,3,4].
Hematological malignancies may manifest as tumor masses in extramedullary sites, which frequently pose a major diagnostic challenge [5]. The myeloid sarcoma (MS) is a rare disease in which one or more tumors of myeloid progenitor cells occur in extramedullary location [3,6]. The MS might affect the ear in a few cases, and mimic other diseases [3,7,8].
Case Report
An 18-year-old male patient, in remission of acute myeloid leukemia subtype M3 (AML-M3) for 9 months, was referred to the Otorhinolaryngology department of a tertiary university hospital due to a one-month progressive right-sided otalgia, fever and purulent otorrhea. He had been unsuccessfully treated with amoxicillin and azithromycin. He also reported right ear fullness within the past 3 months, also unresolved with previous treatments including ear irrigation.
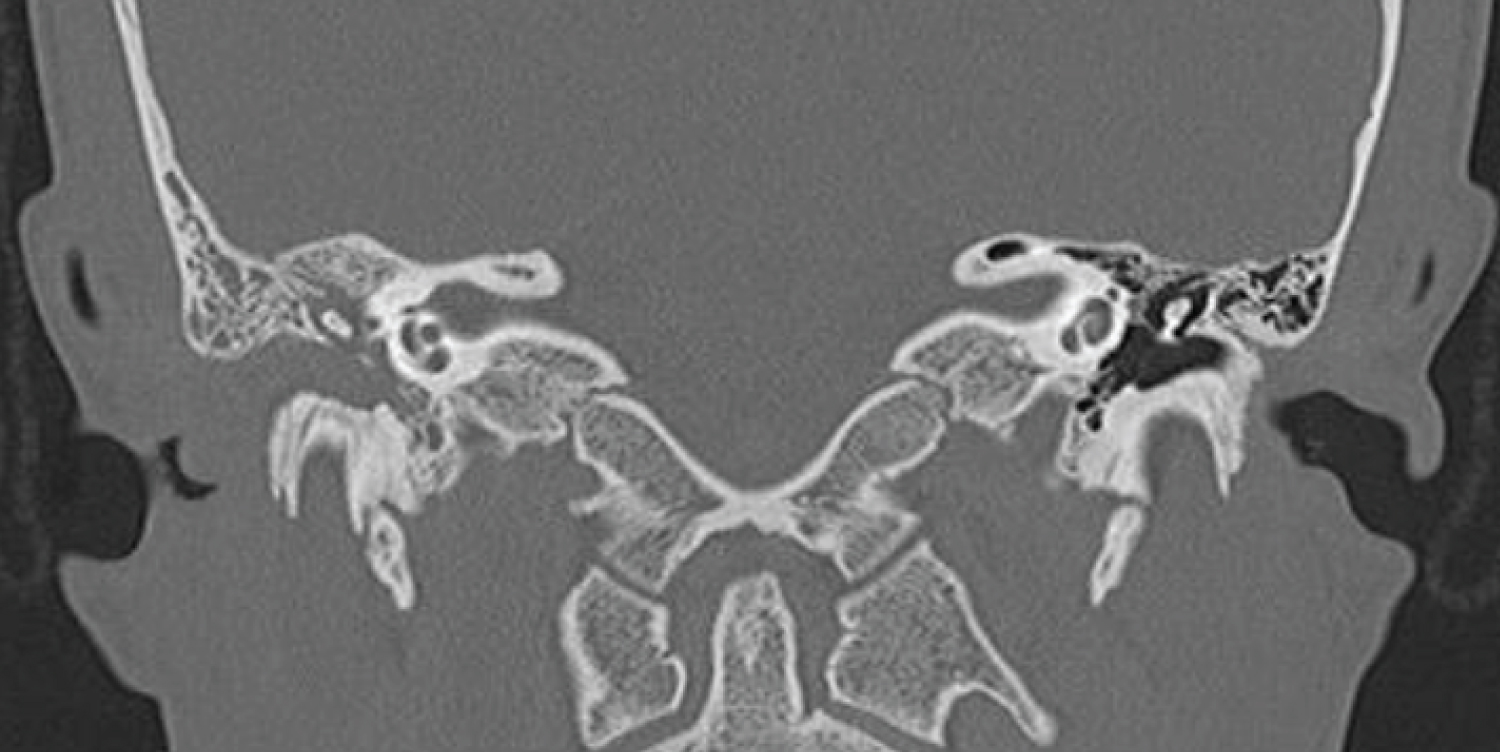
On admission, his physical examination showed submandibular, anterior cervical and pre-auricular lymph nodes on the right side. The nodes were mobile, firm and slightly tender, the largest one measuring 1.5 × 1.5 cm. On otoscopy, an obstructive EAC mass was clearly seen. It had a polypoid aspect with surrounding purulent material. Laboratory evaluation at this point showed no infectious or hematological abnormalities. Computerized tomography (CT) of the neck and temporal bones were performed, revealing an enhancing soft tissue mass on the right EAC. There were no signs of bone erosion and despite adequate pneumatization of mastoid cells, they were found to be obliterated (Figure 1). Right and upper cervical lymph nodes, with a non-specific and probably reactive appearance were seen.
After three days of medical therapy with ceftriaxone 2 g/day and anti-inflammatories, the patient showed good clinical response and normal laboratory tests (hemoglobin 15.9 g/dL; total leukocytes 10,000/mm3; rods 3%; platelets 116,000/mm3; lactate dehydrogenase 184 U/L; C-reactive protein 0.5 mg/L), even though the obstructive mass within the ear canal persisted. The patient was discharged home with Amoxicillin-Clavulanate 500/125 mg every 8 hours for seven more days and a plan of biopsy of both the cervical lymph node and the EAC.
While waiting for the procedure, the patient was readmitted to the emergency department due to macroscopic hematuria and spontaneous ecchymosis. In addition, he reported a progressive enlargement of the lymph nodes previously observed. On otoscopy, the polypoid lesion remained unchanged. His laboratory evaluation at this moment showed: hemoglobin 17.2 g/dL, lactate dehydrogenase 320 U/L, thrombocytopenia (30,000/mm3), and presence of 10% promyelocytes in peripheral blood - an indicative of AML-M3 molecular relapse. Transretinoic acid (ATRA) was promptly initiated. The patient underwent a bone marrow aspiration, whose immunophenotypic analysis revealed proliferation of myeloid lineage, similar to that of the previous diagnosis, determining the relapse of the disease. Rescue therapy with cytarabine, etoposide and ATRA was then initiated.
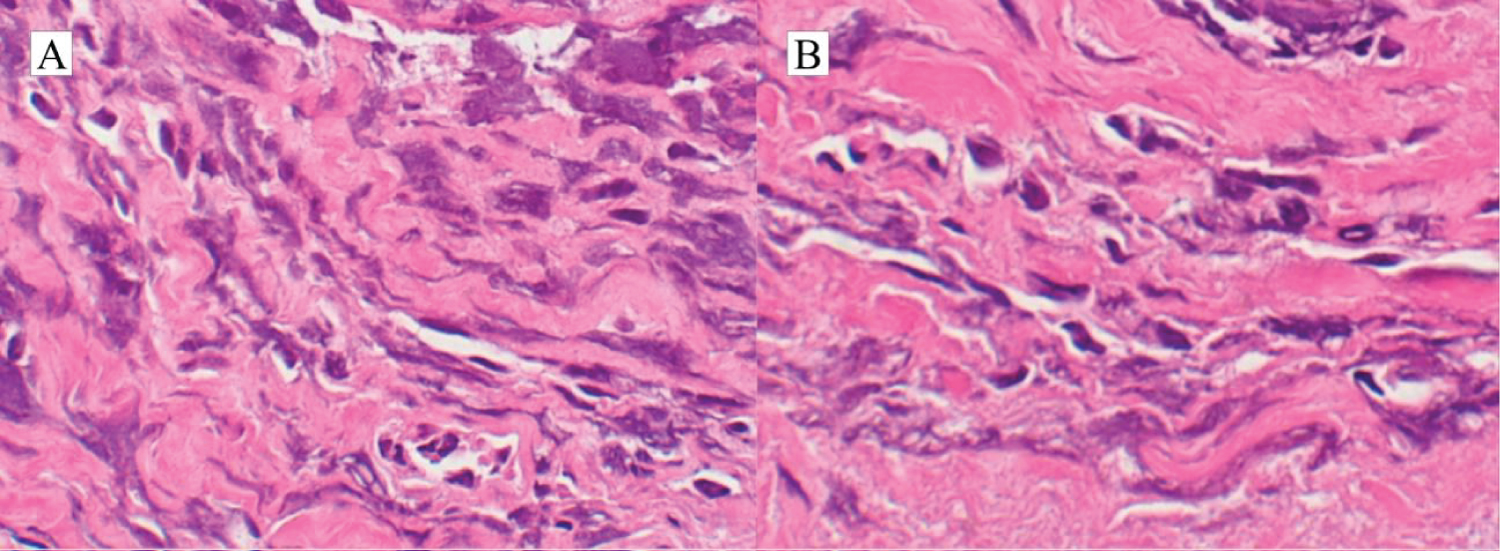
A biopsy of the mass within the right EAC was performed and revealed fibroconnective tissue with proliferation of small and irregular atypical cells with lymphoid morphology (Figure 2). This result gave support to the diagnosis of a myeloid sarcoma and, therefore, the extramedullary relapse. One week later, the patient evolved with clinical regression of the cervical lymph nodes. During his in-hospital treatment, audiometry was performed to assess sudden complaints of hearing loss and dizziness, which showed anacusis on the right ear.
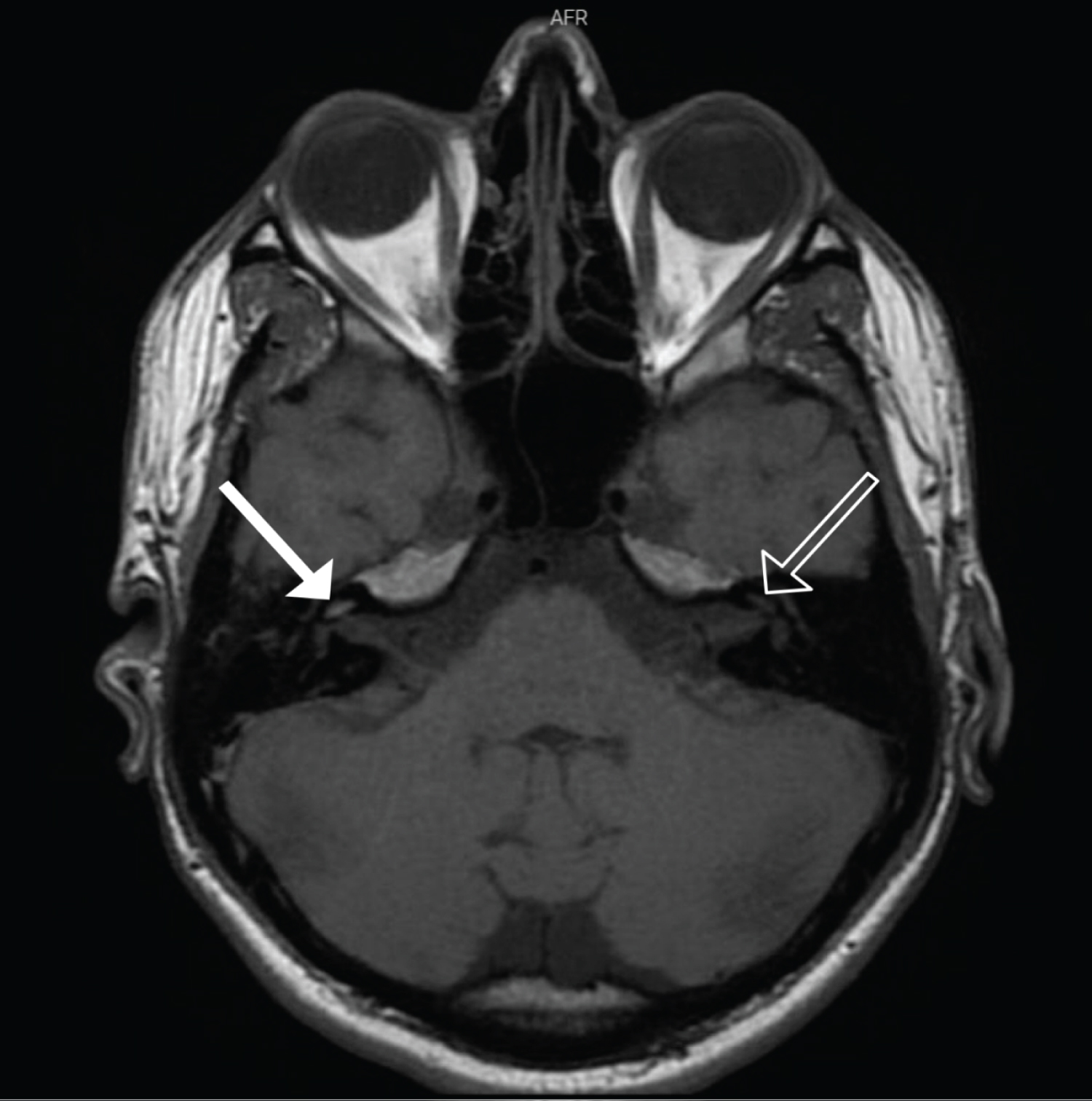
Inner ear magnetic resonance imaging (MRI) revealed a cochlear hyperintensity on T1-weighted images, inferring the presence of methemoglobin and reinforcing the hypothesis of hemorrhagic labyrinthitis (Figure 3).
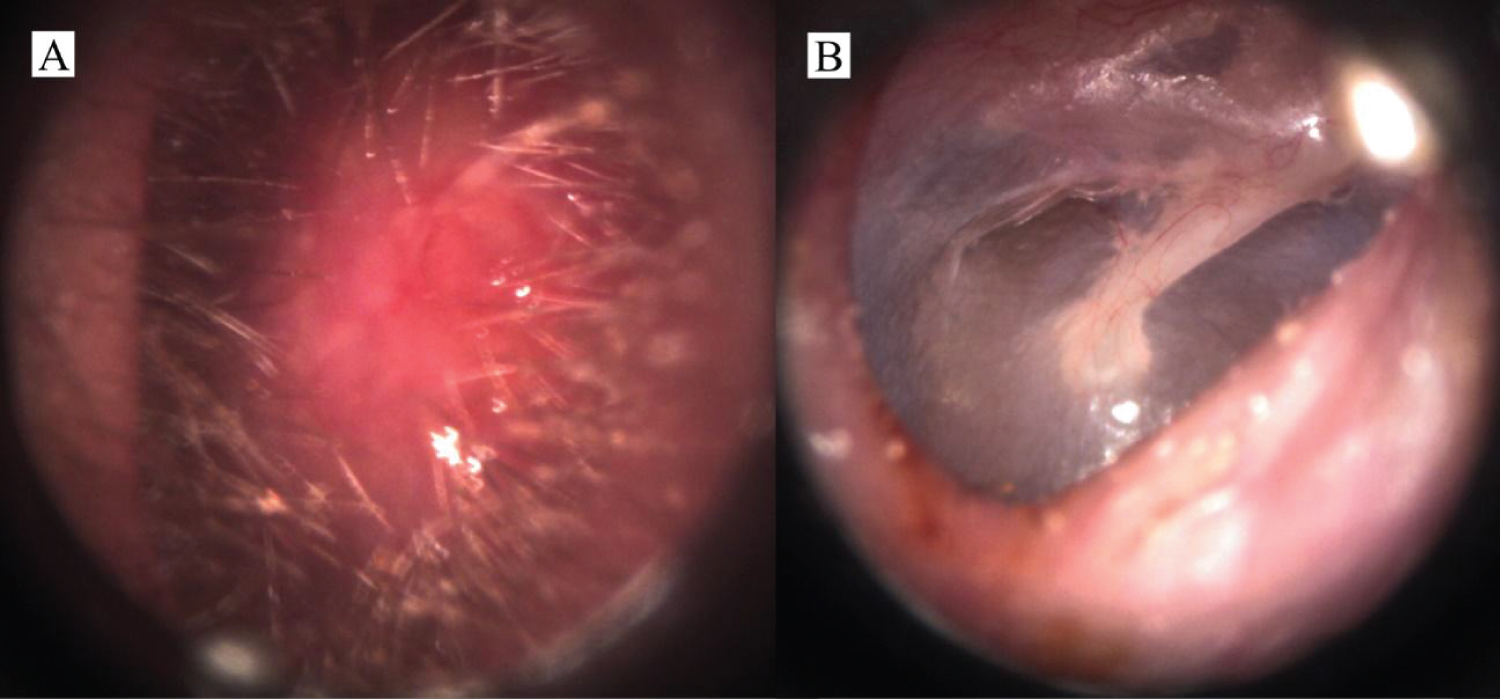
One month after, a significant volumetric reduction of the EAC mass was seen, with only 10% of obstruction, in addition to rapid resolution of the ear pain and local discharge. After treatment completion, total mass regression was documented (Figure 4A and Figure 4B), even though the patientevolved with only mild improvement of the hearing impairment.
Discussion
MS was first described by Burns in 1811. It is also called granulocytic sarcoma, myeloblastoma or chloroma; this last term was coined by Kings, in 1853, due to the greenish color of the lesion attributed to the enzyme myeloperoxidase (MPO) [9].
MS can occur in different moments during the course of myeloproliferative and myelodysplastic diseases [6,10,11]. In some cases, it may be the first manifestation of acute myeloid leukemia or precede it in months to years [6,11]. In others, it may manifest as a first sign of relapse of the disease. MS can affect any tissue and the involvement of multiple anatomical sites is observed in about 10% of the cases [6]. The main sites of occurrence are the skin, lymph nodes, testicles and bone [4,6,12]. Head and neck involvement is uncommon. However, cases of ocular, nasopharynx and central nervous system MS have been reported [7]. As for the ear involvement, it has been rarely reported in the literature.
The overall incidence is estimated at 2.5 to 9.1% among AML patients, with a slight predilection among male individuals. It may be seen at any age, but the highest incidence occurs in pediatric patients [6,7]. In general, the presence of MS in patients with AML, myelodysplasia or myeloproliferative syndromes carries an unfavorable overall prognosis [13].
The spectrum of clinical presentation varies and depends on the location of the tumor. Atypical cases with otological involvement might lead to ear fullness and pain, EAC edema, acute mastoiditis, vertigo, facial paralysis and hearing loss. As the MS affect the temporal bone, it might mimic otomastoiditis, EAC cholesteatoma, acute external otitis, or even necrotizing otitis externa and lymphoma [3,8]. The presence of sensorineural hearing loss or concomitant vertigo might point towards to the concomitant involvement of the inner ear. One might hypothesize that it would be the result from local hemorrhagic-ischemic phenomena or secondary to the action of toxic substances on the cochleovestibular epithelium. This plethora of possible symptoms and differential diagnoses make the recognition of MS challenging. Bilateral MS has been described by Chen, et al. [7] and might pose particular diagnostic difficulties [7,8].
In cases of external otitis with atypical presentation or failure to respond to initial treatment, further investigation with imaging is recommended. With the use of computed tomography scans and/or magnetic resonance imaging, features such as bone erosions, involvement of neurovascular structures and intracranial extension might raise suspicion for malignancies [2,7].
In 2016, Laurindo, et al. found the use of mononuclear leukocytes scintigraphy marked with Tecnecium99m of great value in a patient with extramedullary relapse of AML mimicking necrotizing external otitis [3]. In their case, the laboratory and radiological evaluations led to an initial diagnosis of infection, while the negative scintigraphy pointed out to a neoplasm. Biopsies confirmed the suspicion and therefore seem to be crucial in similar situations.
Even though the presenting symptoms might suggest a much more frequent disease, patients with poor evolution and/or unsatisfactory response to treatment should always be investigated for more serious conditions, including malignancies. Bearing a higher index of suspicion is paramount in cases of patients with a history of cancer or immunosuppression.
The presence of atypical cell infiltrates in any anatomical region with a history of myeloid neoplasm should lead to the suspicion of MS [1]. However, it is often difficult to establish the histopathological diagnosis of leukemic etiology in otological diseases. In fact, it is common to have either insufficient material or significant artefacts in the biopsy specimens [7,14,15]. Histopathology, therefore, is not always satisfactory for the complete identification of the lesion, and can lead to misdiagnosis, even in well-differentiated tumors [15].
Thus, immunophenotyping plays an important role in the diagnosis of extramedullary myeloid tumors [6,15,16]. Positive staining with CD3, CD43 and MPO favors the diagnosis of MS [4,6]. In the present case, the proliferation of undifferentiated cells within the sample was essential to confirm the extramedullary recurrence of AML, and guide prompt chemotherapy. Immunophenotypic analysis was only able to show CD34+ in vascular endothelial cells of the mass, probably due to crushing artifacts within the biopsy material. The dramatic clinical improvement soon after the start of appropriate treatment was compatible with the diagnosis.
In this case, the patient also evolved with sudden neurosensorial hearing loss associated with dizziness due to labyrinthine hemorrhage. Those symptoms in the presence of a T1 hyperintensity on fat-suppressed MRI images within the labyrinth strongly suggest this rare complication of vascular etiology in patients that have hematological disease [17]. The exceptional association of both rare complications that was seen in this report emphasizes the importance of a complete oriented investigation of otological complaints in patients with an onco-hematological disease background.
Conclusion
The differential diagnosis of masses within the EAC can be challenging. MS is a rare condition, which may present a broad range of signs and symptoms. It may rarely affect the ear and mimic other local diseases of much higher prevalence. It should always be considered in patients with history of onco-hematological disease and in those with refractory otologic complaints despite initial management. Further investigation with imaging and biopsies is fundamental. Early diagnosis is an important prognostic factor in such cases and the treatment of the malignancy often results in an abrupt improvement also in the secondary otologic condition.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declarations of Interest
The authors declare no conflicts of interest.
References
- White RD, Ananthakrishnan G, McKean SA, et al. (2012) Masses and disease entities of the external auditory canal: Radiological and clinical correlation. Clin Radiol 67: 172-181.
- Sari S, Battal B, Akgun V, et al. (2015) Metastasis of breast carcinoma to the external auditory canal: Report of an unusual case and literature review. J Belgian Soc Radiol 99: 53-57.
- Laurindo R, Souza S, Moura J, et al. (2016) Extramedullary relapse of acute myeloid leukemia mimicking a necrotizing external otitis: could mononuclear leukocyte scintigraphy be the best diagnostic method? Braz J Otorhinolaryngol 82: 487-490.
- Szomor Á, Baranyai F, Tornóczky T, et al. (2002) Penile chloroma in a patient with secondary acute myeloid leukemia. Eur J Haematol 68: 322.
- Almond LM, Charalampakis M, Ford SJ, et al. (2017) Myeloid Sarcoma: Presentation, Diagnosis, and Treatment. Clin Lymphoma, Myeloma Leuk 17: 263-267.
- Pileri SA, Ascani S, Cox MC, et al. (2007) Myeloid sarcoma: Clinico-pathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia 21: 340-350.
- Chen SM, Silverman DA, Jatana KR, et al. (2019) Myeloid sarcoma presenting as bilateral acute otitis externa in newly diagnosed acute myeloid leukemia. Int J Pediatr Otorhinolaryngol 122: 126-129.
- Mozafari R, Alimoghaddam K, Sotoude H, et al. (2018) A lateral neck myeloid sarcoma presenting as acute otitis externa. Arch Iran Med 21: 544-546.
- Kawatra M, Malukani K and Nandedkar S (2013) Myeloid sarcoma de novo presenting as generalized lymphadenopathy. Clin Cancer Investig J 2: 86.
- Neiman RS, Barcos M, Berard C, et al. (1981) Granulocytic sarcoma: A clinicopathologic study of 61 biopsied cases. Cancer 48: 1426-1437.
- Ascani S, Piccaluga PP and Pileri SA (2003) Granulocytic sarcoma of main biliary ducts. Br J Haematol 121: 534.
- Maeng H, Cheong JW, Lee ST, et al. (2004) Isolated extramedullary relapse of acute myelogenous leukemia as a uterine granulocytic sarcoma in an allogeneic hematopoietic stem cell transplantation recipient. Yonsei Med J 45: 330-333.
- Jaffe ES, Organization WH (2001) Cancer IA for R on Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. IARC Press.
- Paparella MM, Berlinger NT, Oda M, et al. (1973) Otological manifestations of leukemia. Laryngoscope 83: 1510-1526.
- Menasce LP, Banerjee SS, Beckett E, et al. (1999) Extra-medullary myeloid tumour (Granulocytic sarcoma) is often misdiagnosed: A study of 26 cases. Histopathology 34: 391-398.
- Chen J, Yanuck RR, Abbondanzo SL, et al. (2001) c-Kit (CD117) reactivity in extramedullary myeloid tumor/granulocytic sarcoma. Arch Pathol Lab Med 125: 1448-1452.
- Salomone R, Abu TAA, Chaves AG, et al. (2008) Sudden hearing loss caused by labyrinthine hemorrhage. Braz J Otorhinolaryngol 74: 776-779.
Corresponding Author
Nicole Tássia Amadeu, Complexo Hospital de Clínicas - Universidade Federal do Paraná (CHC-UFPR), R. Gen. Carneiro, 181 - Alto da Glória, Curitiba, Paraná, 80060-900, Brazil, Tel: +55-41-995057025; +55-41-33606588.
Copyright
© 2022 Mangia LRL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.