Sub-Acute Brachial Plexopathy After Mid-Shaft Clavicle Fracture
Abstract
Case
A 52-year-old woman sustained a closed, mid-shaft, displaced right clavicle fracture with subsequent brachial plexopathy. Examination revealed weakness in deltoid, as well as with elbow, wrist, and finger extension, with diminished sensation. Radiographs revealed shortening and displacement. CT revealed a Z-fragment. EMG revealed a posterior cord brachial plexus injury. The patient initially declined surgery, but after 1 month without resolution of symptoms, she agreed to surgery. Intraoperatively, meticulous microscopic neurolysis was performed and the fracture was reduced and fixed using plate and screws. The patient gradually recovered nerve function postoperatively.
Conclusion
Although brachial plexopathy with clavicle fractures are rare, operative intervention and careful attention to monitoring the neurologic status of recovering patients can improve functional outcome.
Introduction
Brachial plexus palsy after clavicle fracture is rare in both adult and pediatric populations, with a reported incidence of under 1%, and variable outcomes despite operative intervention [1,2]. They are typically caused acutely by direct fragment compression, which although exceedingly rare have been reported in the context of high energy mechanisms, such as motorcycle accidents, with potentially fatal associated vascular disruption [3-6]. In adults, non-union, mal-union, hypertrophic callous, and pseudoaneurysm of the subclavicular vessels are responsible for brachial plexus injuries, which can occur months to years after injury [7-9]. This article presents the case of a patient with subacute brachial plexus palsy after clavicle fracture.
The authors have obtained the patient's informed written consent for print and electronic publication of the case report.
Case Report
A 52-year-old, left-hand dominant woman sustained a closed right mid-shaft clavicle fracture and closed left distal radius fracture after slipping on a rock while walking on the beach (Figure 1). She had a history of hypothyroidism, worked as a registered nurse in a rehabilitation hospital, and was a non-smoker. Her neurologic exam and pulse were intact and documented on initial examination. The fracture demonstrated more than two centimeters of displacement, and the patient was offered the option of surgical intervention, but she declined this and asked to be treated non-operatively. The patient progressed well, with improving pain, however, she returned to clinic 2 weeks after her injury complaining of a sudden loss of function in her right upper extremity earlier that day, as well as numbness and a cold sensation throughout her extremity.
Physical examination revealed deficits in the posterior cord of brachial plexus distribution. She could not extend her elbow actively, extend her wrist, or extend her thumb/digits; she also had weakness with deltoid muscle testing. She had decreased sensation of the deltoid and dorsum of the forearm. A computed tomography (CT) was obtained to better characterize the fracture pattern, which revealed displacement, shortening, and a Z-fragment (Figure 2); no pseudoaneurysm was present. An electromyogram (EMG) revealed prolonged latency and reduced amplitude in the axillary nerve motor responses of the deltoid. The same was true of radial motor responses, including the triceps, extensor carpi radialis longus, extensor carpi radialis brevis, brachioradialis, extensor pollicis longus, extensor pollicis brevis, abductor pollicis longus, extensor pollicis et indicis communis, extensor digitorum communis, extensor digiti minimi, and extensor carpi ulnaris. These findings suggested a right brachial plexus palsy involving its posterior cord. The patient was encouraged to undergo surgical intervention to attempt reduction and fixation of her clavicle fracture and to treat her plexopathy, but she continued to decline, citing fear of the surgical procedure and of undergoing a general anesthetic. After 1 month she returned to clinic with no improvement in her function, and accepted surgical intervention.
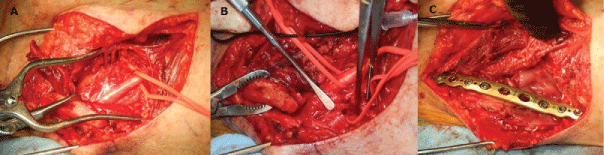
A standard approach to the clavicle was used. During surgery, the brachial plexus appeared pale and was encased in fibrous scar tissue attached to the lateral fracture fragment. With the assistance of a hand surgeon, microscopic neurolysis was performed, and the cords were wrapped in an amniotic membrane to prevent scarring. The clavicle fracture was reduced and fixed using a metal plate and screws figure 3, placed anteriorly. Morselized allograft cancellous bone graft was used at the fracture site. Over the next 6 months, the patient's symptoms improved significantly, with gradual resolution of her numbness and improvement in the function of her extremity (Figure 4). By 8 months post-op, the patient was able to resume her career as an occupational therapist, hobbies such as yoga and cooking, and her activities of daily living with little difficulty. By one year after surgery, her range of motion improved from 70 to 160° of forward flexion, from 60 to 160° of abduction, from neutral to 40° of external rotation, and internal rotation from her buttocks to T12. Her post-operative Single Assessment Numeric Evaluation (SANE) score was 90, Disabilities of the Arm, Shoulder, and Hand (DASH) score was 7.41, American Shoulder and Elbow Surgeons (ASES) score was 80, Visual Analog Scale (VAS) was 3, and Constant score was 78.15. Her sensation was intact throughout the extremity, and there were no motor deficits noted.
Discussion
The vast majority of clavicle fractures occur in the context of low energy mechanisms, for which brachial plexus injuries are exceedingly rare [1,3]. Although recent research suggests that significant shortening or displacement of the clavicle can lead to functional deficits in the associated extremity, conservative treatment of both displaced and nondisplaced clavicle fractures has been an accepted treatment option, documented to produce satisfactory outcomes [2,10].
Initial surgical treatment of mid-shaft clavicle fractures is increasing [11]. Indications for surgical intervention include: open fractures, nonunion or cases where nonunion is likely, severe comminution or angulation of the fracture, with little bony contact, inability to tolerate conservative treatment, and neurovascular compromise [3,5].
Acute brachial plexus injuries in the context of mid-shaft clavicle fractures can be the result of direct compression by the clavicle (neurogenic thoracic outlet syndrome) or by high-energy traction injuries, where the medial end of the distal clavicular fragment is pulled inferiorly by the weight of the arm and medially by the latissimus dorsi and pectoralis major muscles to impinge on the brachial plexus [1,5,12]. Despite the anatomic protection of the sternocleidomastoid muscle, which pulls the proximal fragment upward, and the subclavius muscle and clavipectoral fascia between the clavicle and neurovascular structures, the close proximity of the brachial plexus and axillary artery as it passes between the anterior and middle scalene muscles prior to crossing the lateral border of the first rib creates the risk for both traction and compression. This risk is heightened in fractures of the mid-shaft of the clavicle due to the loss of stability conferred by the stabilizing ligaments both proximally and distally [1,3]. Displacement at the fracture site is also increased by scapular ptosis, which pulls the fractured ends of the clavicle closer to the brachial plexus, and by protraction of the scapula [13].
In the acute setting, clavicle fracture-related brachial plexus injuries are typically supraclavicular lesions when caused by traction injuries, and typically operative fixation of the clavicle does not affect the neurological outcome [9]. In the case of infraclavicular lesions caused by external compression, such as hematoma, or bony fragment, prognosis is favorable if prompt surgical intervention is performed [4,5].
Chronic brachial plexus injuries have been reported in the months after injury, yet some injuries have been reported occurring in only a few weeks after injury. All of these subacute injuries have been in the context of pseudoaneurysm of the subclavicular artery and vein, while more chronic injuries have occurred in the context of non-union, mal-union, and hypertrophic callous [7-9]. In the case of our patient, her brachial plexus injury occurred only 2 weeks after injury, and in the 1 month time period that she declined operative intervention, her plexus significantly scarred in, without any evidence for pseudoaneurysm.
A number of factors make brachial plexus injury onset and recovery difficult to predict. The anatomic variability on the costoclavicular space between patients contributes to this [7]. Our patient was very lean, with very little soft tissue protection, possibly contributing to the development of her palsy. There remains the question of whether sling and swathe is too tight a bandage or contributes to posttraumatic edema, both of which have been documented in cases of post-injury brachial plexus palsy [14]. Our patient initially used a sling and swathe, without any initial neurologic symptoms, yet suddenly developed neurologic symptoms at 2 weeks from injury. In described cases where neurologic deficits were delayed yet immediate, poorer outcomes were reported compared to those that had a gradual onset of symptoms [7]. Persistent symptoms may include pain, dysesthesia, and cold intolerance [15]. A specific neurologic exam can be difficult to obtain due to pain that may be disguised as weakness, as in the case of deltoid weakness during physical examination of our patient. Thankfully, in the case of our patient, she fully recovered from her brachial plexus injury despite the delayed yet immediate onset of her symptoms and despite the delay from discovery of her nerve injury to operative intervention. Our patient had an injury to her posterior cord of brachial plexus, which is consistent with current literature, where compression of the medial and posterior cords has been reported most frequently [1].
Patients with clavicle fracture who develop brachial plexus injuries should be offered immediate surgical intervention, with careful pre-operative assessment of clinical signs and symptoms. Studies have suggested that T1-weighted magnetic resonance imaging (MRI) obtained in the sagittal plane is a reliable method for detecting brachial plexus compression, and EMG study can help deduce with cords are involved in the injury [3,16]. MR neurography is another useful high-resolution imaging technique, in particular those provided by 3 Tesla equipment, to distinguish a normal plexus from a pathological one. They can accurately determine the location of a lesion (root, trunk, division or cord), and whether the lesion affects multiple areas [17]. Our patient had initially declined surgical intervention, despite CT findings of a Z-fragment in the context of a significantly shortened and displaced fracture pattern; her EMG revealed a clear posterior cord injury, but the patient initially had fears about undergoing an operation.
Surgical intervention should include care when dissecting near the brachial plexus. Performing neurolysis, clearing any areas of tension on the cords by removing any bony fragments or callus that are suspected of causing nerve compression is not without risk. Amniotic nerve wraps have recently been approved by the Food and Drug Administration (FDA) and demonstrated in literature to improve the histological and electrophysiologic outcomes after neuroloysis and nerve injury repair via decreasing scar formation around the nerves [18,19]. The goal is to create a barrier to prevent scarring between the bleeding ends at the fracture site and overlying plate from the nerves that have been dissected free from the callus. In our patient, amniotic nerve wraps were utilized after neuroloysis.
Rigid fixation, either by plate-and-screw fixation, or by intramedullary (IM) rod fixation, are recommended to prevent redisplacement and decrease the risk of further nerve damage, although IM rods have been shown to increase soft tissue tension during reaming [3,9]. For these reasons, we used a compression plate-and-screw fixation technique after careful neurolysis. Anterior plating is the technique of choice for the authors to decrease plate prominence in thin individuals and to decrease the risk of perforation of neurovascular structures during fixation.
Conclusion
This case described the rare but deleterious risk of neurologic injury in mid-shaft clavicle fractures, highlighting the importance of documenting neurovascular status and frequent follow-up during the healing process, as well as counseling of patients to be aware of developing signs and symptoms. It also shows how delayed intervention for acute brachial plexus injuries can result in good outcomes; immediate intervention remains the preferred treatment choice to increase chances of a good outcome.
References
- I Gill, J Quayle, M Fox (2013) A low energy pediatric clavicle fracture associated with acute brachial plexus injury and subclavian artery compression. Ann R Coll Surg Engl 95: e30-e33.
- Canadian Orthopaedic Trauma Society (2007) Nonoperative treatment compared with plate fixation of displaced midshaft clavicular fractures. A multicenter, randomized clinical trial. J Bone Joint Surg Am 89: 1-10.
- Lin CC, Lin J (2009) Brachial plexus palsy caused by secondary fracture displacement in a patient with closed clavicle fracture. Orthopedics 32.
- Barbier O, Malghem J, Delaere O, et al. (1997) Injury to the brachial plexus by a fragment of bone after fracture of the clavicle. J Bone Joint Surg Br 79: 534-536.
- Chen DJ, Chuang DC, Wei FC (2002) Unusual thoracic outlet syndrome secondary to fractured clavicle. J Trauma 52: 393-398.
- Derham C, Varghese M, Deacon P, et al. (2007) Brachial plexus palsy secondary to clavicular nonunion. J Trauma 63: E105-E107.
- Howard FM, Shafer SJ (1965) Injuries to the clavicle with neurovascular complications: A study of fourteen cases. J Bone Joint Surg Am 47: 1335-1346.
- Watanabe K, Matsumura T (2005) Late-onset brachial plexus paresis caused by subclavian pseudoaneurysm formation after clavicular fracture. J Trauma 58: 1073-1074.
- Rumball KM, Da Silva VF, Preston DN, et al. (1991) Brachial-plexus injury after clavicular fracture: case report and literature review. Can J Surg 34: 264-266.
- Nowak J, Holgersson M, Larsson S (2005) Sequelae from clavicular fractures are common: a prospective study of 222 patients. Acta Orthop 76: 496-502.
- Murray IR, Foster CJ, Eros A, et al. (2013) Risk factors for nonunion after nonoperative treatment of displaced midshaft fractures of the clavicle. J Bone Joint Surg Am 95: 1153-1158.
- Konkin D, Schubert H (2000) Emergency case. Clavicle fracture. Can Fam Physician 46: 2201-2203.
- Ristevski B, Hall JA, Pearce D, et al. (2013) The radiographic quantification of scapular malalignment after malunion of displaced clavicular shaft fractures. J Shoulder Elbow Surg 22: 240-246.
- Miller DS, Boswick JA Jr (1969) Lesions of the brachial plexus associated with fractures of the clavicle. Clin Orthop Relat Res 64: 144-149.
- Kitsis CK, Marino AJ, Krikler SJ, et al. (2003) Late complications following clavicular fractures and their operative management. Injury 34: 69-74.
- Demondion X, Boutry N, Drizenko A, et al. (2000) Thoracic outlet: anatomic correlation with MR imaging. AJR Am J Roentgenol 175: 417-422.
- Cejas C, Rollán C, Michelin G, et al. (2016) High resolution neurography of the brachial plexus by 3 Tesla magnetic resonance imaging. Radiologia 58: 88-100.
- Kehoe S, Zhang XF, Boyd D (2012) FDA approved guidance conduits and wraps for peripheral nerve injury: a review of materials and efficacy. Injury 43: 553-572.
- Henry FP, Goyal NA, David WS, et al. (2009) Improving electrophysiologic and histologic outcomes by photochemically sealing amnion to the peripheral nerve repair site. Surgery 145: 313-321.
Corresponding Author
Mark Schultzel, Southern California Permanente Medical Group, San Diego, California, USA.
Copyright
© 2017 Schultzel M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.