Paraspinal Metastasizing Pleomorphic Adenoma
Abstract
Pleomorphic adenomas (benign mixed tumors) are the most common benign tumors arising in the salivary glands. Rarely, they can undergo malignant transformation (carcinoma ex pleomorphic adenoma) or metastasize via hematogenous or lymphatic spread. We describe a case of a 51-year-old male with a benign metastasizing pleomorphic adenoma of the vertebrae and epidural space of the lumbar spine who presented with lower back pain. He developed spinal metastases within 8 months of excision of his primary tumor, which arose in the right submandibular gland. Most recently, he went on to develop pulmonary metastases about a year after surgical excision of the spinal metastases. We review the literature on benign metastasizing mixed tumors which involve the spine.
Keywords
Benign metastasizing pleomorphic adenoma, Benign mixed tumor, Spine tumor, Salivary gland tumor
Introduction
Pleomorphic adenomas (PA), or benign mixed tumors, account for 70-80% of benign tumors of the salivary glands in adults [1]. The vast majority of them (84%) arise in the parotid glands; 8% arise in the submandibular glands, and the remaining 8% occur in the sublingual and minor salivary glands [1]. Rarely, malignant transformation (e.g. carcinoma ex pleomorphic adenoma) of these benign tumors can occur. Furthermore, there are 80 case reports, between 1942-2014, which describe metastases of benign PAs without pathologic evidence of malignant transformation [2]. The predominant primary lesion in these benign metastatic PAs is the parotid gland (74.1%), followed by the submandibular gland (14.8%); palatal, nasal, lingual, and pulmonary sources comprise the remaining 10.1% [2]. The most likely sites of metastasis are bone (36.6%), lung (33.8%), and cervical lymph nodes (20.1%) [2], with time between primary diagnosis and presentation of metastases varying widely (mean 14.9 years, range 0-51 years) [2]. Involvement of the spine is extraordinarily rare. Herein, we present a case of a 51-year-old male whose submandibular PA metastasized to his lumbar spine and epidural space within a year of having it removed. We review the literature on spinal involvement by this lesion.
Case Report
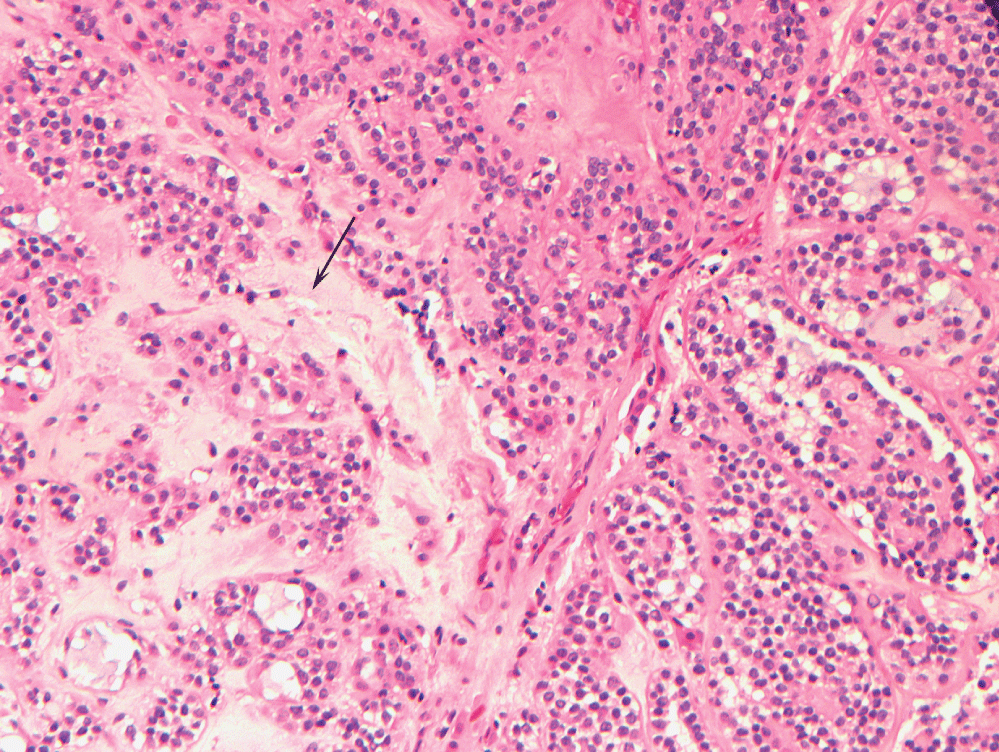
A 51-year-old male with a 40 pack year history of smoking, diabetes type 2, hypertension, hyperlipidemia, and diastolic congestive heart failure presented with a right submandibular mass and no other symptoms. Fine needle aspiration of the mass showed atypical cells and low grade epithelial neoplasm; in conjunction with computed tomography (CT) findings, the presumptive diagnosis was pleomorphic adenoma of the submandibular gland. The patient opted for conservative management until four years later, at which time he underwent resection of the tumor which had moderately increased in size. The excised mass measured 2.9 × 3.3 × 3.9 cm. The tumor was marked by a mixture of epithelial and myoepithelial cells arranged against a chondro-myxoid-like matrix, consistent with a pleomorphic adenoma (Figure 1).
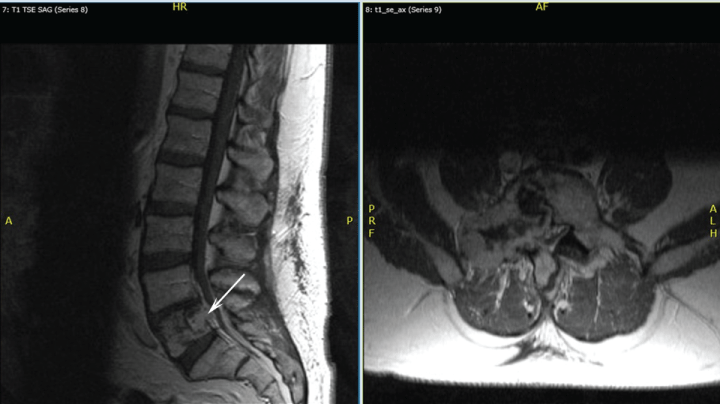
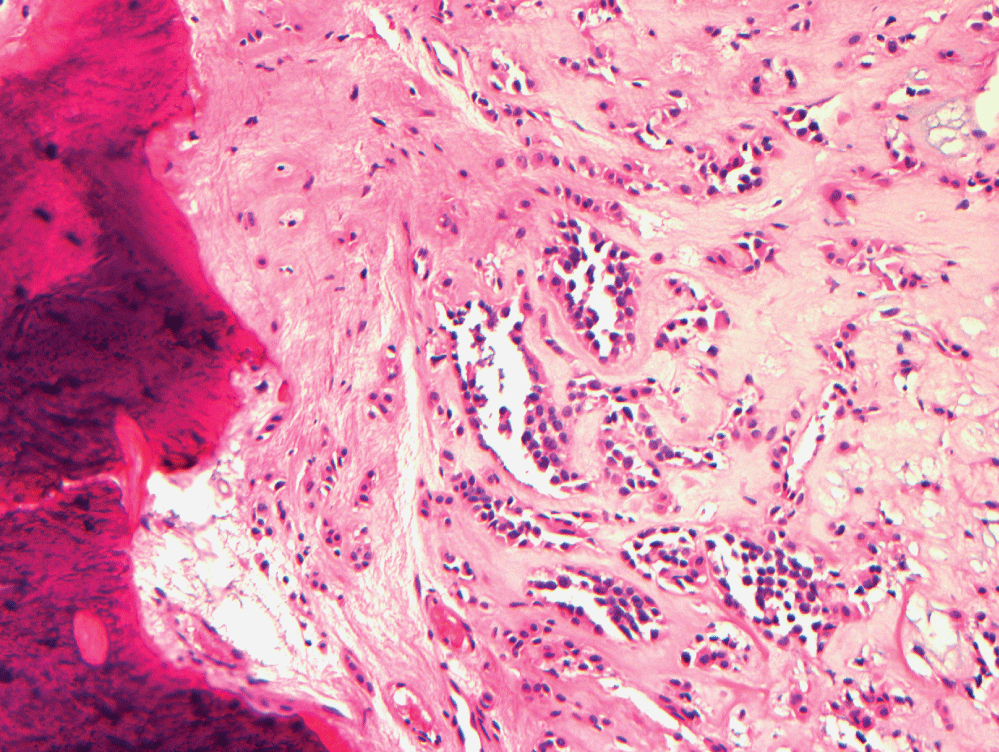
Sixteen months after this excision, prompted by the patient's complaint of eight months of lower back pain without neurologic impairment, magnetic resonance imaging (MRI) studies indicated the presence of a lesion involving his L5 vertebrae. Figure 2 shows MRI findings one year after this diagnosis. A whole body bone scan and positron emission tomography (PET) scan showed increased uptake only at the L5 vertebral body. A CT-guided biopsy of the spinal lesion showed similar pathology to his submandibular gland lesion, confirming the presence of benign metastasizing pleomorphic adenoma (Figure 3). The tumor demonstrated positive staining with antibodies to cytokeratins AE1/3 (1:200 dilution; Millipore - Rocklin, CA) and CAM 5.2 (1:10 dilution; Becton Dickinson - San Antonio, TX). The tumor also demonstrated positive staining with antibodies to S-100 protein (1:800 dilution; DAKO - Carpinteria, CA) and glial fibrillary protein (GFAP) (1:600 dilution; DAKO). The tumor did not stain with antibodies to chromogranin (1:00 dilution; DAKO) and TTF-1 (prediluted; Ventana - Tucson, AZ). Given the location of the lesion, conservative management was the initial approach.
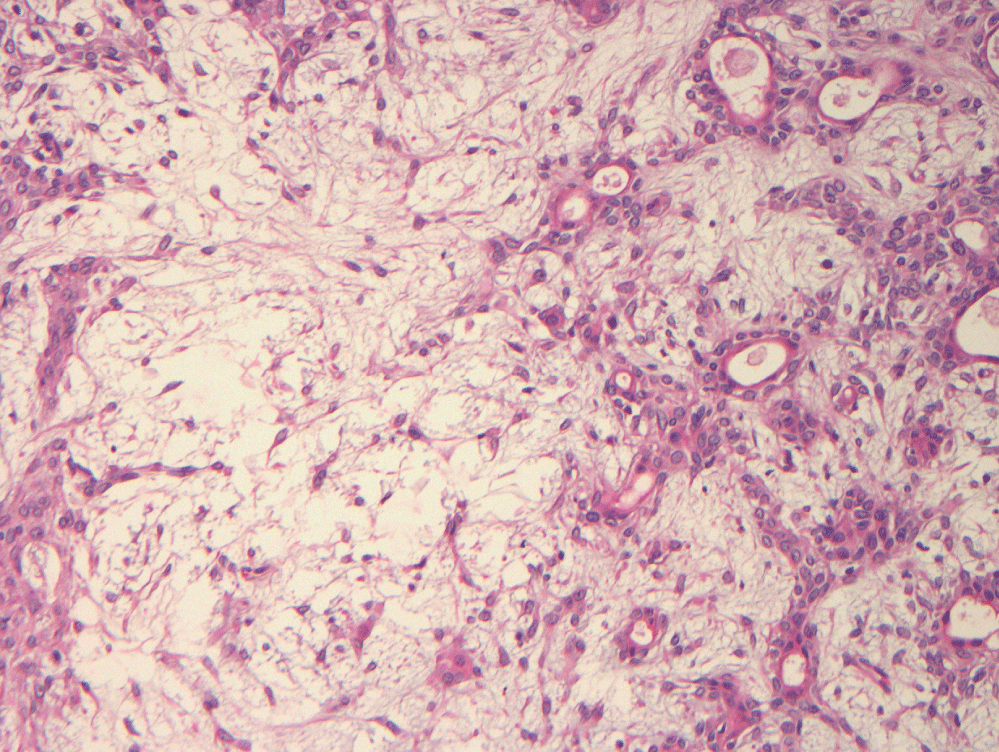
Over the next five months, he experienced worsening pain with physical activity. He underwent one round of palliative stereotactic gamma radiosurgery. His symptoms progressed to include numbness along his right L5 dermatome. His pain and numbness worsened without much alleviation from nerve blocks. Thirty months after the first biopsy of the spinal lesion, he underwent a ventral tumor resection at L5-S1 with decompression of L4-S1 with fusion. Tumor sampled from the epidural region was similar in appearance to the salivary gland neoplasm and the previously biopsied fifth lumbar vertebral mass (Figure 4). He initially experienced complete resolution of his back pain for about six months, at which time, he developed recurrent back pain and tingling of his left foot; for this, he underwent another round of stereotactic radiosurgery at L5 with some improvement of symptoms.
One year after his second spinal surgery, he was found to have a left upper lobe lung lesion and pleural effusion. Biopsy confirmed that the pulmonary lesion was metastatic pleomorphic adenoma. CT imaging of the rest of the body showed no other tumors. His spinal lesion has remained stable in size on follow-up MRI studies, although he developed a right-sided foot drop that is most likely due to his spinal lesion. A recent follow-up CT scan of the chest, done one year after the pulmonary lesions were found, showed multiple pulmonary nodules and small increase in size of previously noted nodules.
Discussion
Typically, metastatic disease occurs in local craniofacial tissues and cervical lymph nodes; however, pulmonary sites are also very common [2-3]. Central nervous system involvement in metastasizing PAs is particularly rare; very few cases in the English literature report intracranial or spinal involvement, especially outside of the cervical spine, [2,4]. In a literature review by Knight, et al. [2], there were eight reports of spinal involvement, and five of these had a submandibular primary site, which is disproportionally high compared to the reported 14.8% incidence of submandibular metastatic PAs relative to other primary PA sites. Furthermore, there were only twelve reports in total of submandibular PAs developing metastatic disease [2]. When compared to three of 60 reported spinal metastatic PAs which originated in the parotid gland, it seems that submandibular PAs, when they rarely metastasize, may have a somewhat higher propensity to metastasize to the spine. Almost half of patients (40.7%) developed multiple sites of metastasis, as was the case with our patient [2]. Accurate survival rates were not able to be determined [2].
Proposed theories as to how these ostensibly benign appearing salivary gland tumors paradoxically metastasize include hematogenous or lymphatic spread by iatrogenic means at the time the primary salivary gland neoplasm is excised; pulmonary metastases have been theorized to be directly aspirated from the primary site [5]. In our patient's case, the spinal lesions were symptomatic and appeared after the salivary gland excision; chronologically, this does support possible iatrogenic spread. His pulmonary lesions were not present until five years after his the primary excision but only one year after the excision of his spinal metastases, which invites the question as to whether his pulmonary nodules resulted from spread from the salivary gland tumor or spinal tumor.
Knight, et al. [2] noted that the average time interval between presentation of primary PA and metastasizing PA drastically decreases with age, with the interval to metastasis being shorter in older individuals. However, the sample size for patients > 40-years-old was only n = 5 and it is not reported when in this course primary excision was performed [2]. Regardless, our patient had a short time course between presentation and metastatic disease, and this trend was even more apparent after the primary excision.
Guidelines regarding the management of metastatic PA outside the head and neck region are unclear due to the small size of previously reported cases, and hence, most of the recommended therapies are based on management strategies of recurrent PA or local metastatic disease. For prevention, Nouraei and colleagues recommended enucleation of PA not be performed due to incomplete clearance of atypical cells which is strongly associated with local recurrence; rather, total surgical excision is preferred [3]. Regarding the options for symptomatic metastasizing PA, total resection is generally favored; however, the role of adjuvant radiotherapy is still under investigation [6].
The spinal tumor required radiotherapy twice, once before his spinal surgery, which reportedly alleviated his symptoms for two weeks, and again after MRI studies showed recurrence post spinal excision. The utility of this therapy is not clear with our case, as imaging studies showed a stable size of the lesion before and after. Our patient is followed with frequent MRI and CT scans for continued surveillance. Nouraei, et al. stressed the importance of follow up via PET/CT scans to monitor for disease recurrence [3].
In conclusion, pleomorphic adenomas arising in the salivary glands may rarely metastasize. In patients presenting with a spinal or paraspinal mass following excision of a salivary gland pleomorphic adenoma, a metastasis should be considered in the differential diagnosis.
References
- Thoeny H (2007) Imaging of salivary gland tumors. Cancer Imaging 7: 52-62.
- Knight J, Ratnasingham K (2015) Metastasizing pleomorphic adenoma: systematic review. Int J Surg 19: 137-145.
- Nouraei S, Ferguson M, Clarke PM, et al. (2006) Metastasizing pleomorphic salivary adenoma. Arch Otolaryngol Head Neck Surg 132: 788-793.
- James Knight, Leslie R Bridges, Pawanjit S Minhas, et al. (2016) Metastasizing pleomorphic adenoma of the submandibular salivary gland presenting with spinal cord compression: case report and review of cases with CNS involvement. Br J Neurosurg 22: 1-3.
- Wermuth DJ, Mann CH, Odere F (1988) Metastasizing pleomorphic adenoma arising in the soft palate. Otolaryngol Head Neck Surg 99: 505-508.
- Witt RL, Eisele DW, Morton RP, et al. (2015) Etiology and management of recurrent parotid pleomorphic adenoma. Laryngoscope 125: 888-893.
Corresponding Author
Richard Prayson, MD, MEd, Department of Anatomic Pathology, L25 Cleveland Clinic, 9500 Euclid Avenue, Cleveland, Ohio 44195, USA, Tel: 216-444-8805, Fax: 216-445-6967.
Copyright
© 2017 Bryant C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.