Tumor Lysis Syndrome in Gastrointestinal Stromal Tumor Treated with the Oral Tyrosine Kinase Inhibitor Imatinib: A Case Report
Abstract
Tumor lysis syndrome in hematological malignancy is more common than in solid tumors. Little literature has been published on tumor lysis syndrome in solid tumors. In this paper we present a case of metastatic gastrointestinal stromal tumor treated with imatinib that results in tumor lysis syndrome and liver failure. Clinicians should be alert about the possibility of tumor lysis syndrome and liver failure in patients with hepatic metastatic gastrointestinal stromal tumor treated with imatinib.
Keywords
Tumor lysis syndrome, Gastrointestinal stromal tumor, Imatinib.
Introduction
Tumor lysis syndrome is a well-known complication of hematologic malignancies, such as in leukemia and lymphoma. However, tumor lysis syndrome is rare but potential mortality in solid tumors, especially advanced metastatic tumor [1]. Tumor lysis syndrome is characterized by rapid cellular lysis causing the release of intracellular contents into the blood, and presented with hyperuricemia, hyperphosphatemia, hypocalcemia, and acute renal failure [2]. The main stays of managements are flourishing hydration and allopurinol for hyperuricemia [1,3].
Gastrointestinal Stromal Tumor (GIST) is a kind of gastrointestinal mesenchymal tumor. GIST arises commonly in the stomach, followed by the small intestine, colon and rectum, mesentery or omentum, and esophagus. Most GISTs have a gain-of-function mutations in the c-KIT proto-oncogene resulting in stimulus for cell growth. Chemotherapy is notoriously unresponsive on GIST [4] and surgery has poor prognosis due to tumor recurrence and metastatic disease [5]. Targeted molecular therapy with oral tyrosine kinase inhibitors (such as imatinib and sunitinib) has a considerable improved outcome [5]. Due to the effective treatment, imatinib was reported to cause mortality of tumor lysis syndrome in a metastatic GIST case [6]. Large friable tumor mass was noted by autopsy and dramatic tumor cells were killed by histology, consistent the potent effect of imatinib [6]. Sunitinib also was reported to cause fetal tumor lysis syndrome with acute renal failure in a metastatic GIST case after the fail of imatinib treatment [7]. Reduced dose of imatinib 300 mg therapy and prevention strategy of aggressive hydration, rasburicase and allopurinol were reported to completely diminish GIST without recurrence of tumor lysis syndrome, induced by imatinib 400 mg [8].
Here, we report a metastatic GIST case with liver metastasis and jaundice that developed tumor lysis syndrome immediately after treatment with the oral tyrosine kinase inhibitor, imatinib.
Case Report
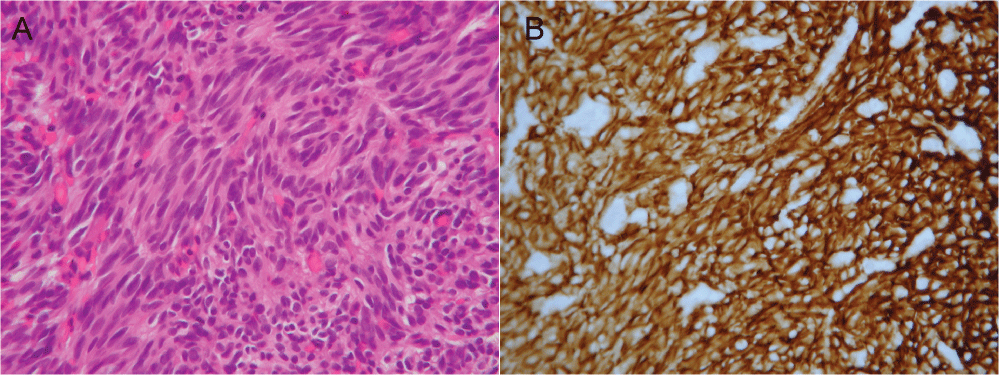
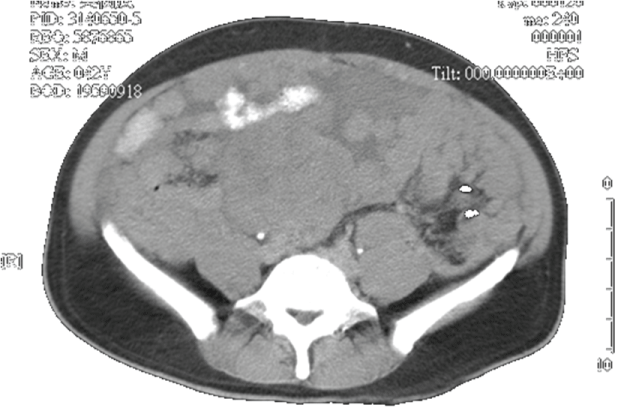
A 43-year-old male patient presented with a protruding mass on the right groin. He was healthy before and had no any systemic disease. Inguinal hernia was initially suspected and he prepared to receive hernioplasty. Laboratory data showed mild elevated total bilirubin, direct bilirubin and alkaline phosphatase (Table 1). However, computerized tomography scan demonstrated multiple confluent masses involving in the liver, mesentery, omentum, and peritoneum (Supplemental Figure 1). A mass in the liver and peritoneal metastasis were found. Sonogram-guide biopsy was performed from the peritoneal mass. The pathology of biopsy disclosed spindle cells with marked hypercellularity and pleomorphism and positive for CD-117 (Figure 1). Metastatic GIST with mild jaundice was diagnosed.
Imatinib 400 mg once daily was prescribed. However, he lost consciousness the next day. His biochemistry data showed hyperkalemia, hyperuricemia, hyperphosphatemia, metabolic acidosis and hyperammonia. These results were compatible with the definition of laboratory tumor lysis syndrome and hepatic encephalopathy. Following acute renal failure was also in accordance with definition of clinical tumor lysis syndrome. Then he was transferred to intensive care unit because of unstable vital signs. His condition progressively went downhill in spite of aggressive management. He died 4 days after receiving his initial dose of imatinib.
Discussion
Surgery remains the primary treatment in localized GIST when the operation can be smoothly carried out without excess morbidity or functional compromise [5]. Chemotherapy and radiotherapy have shown little efficacy in patients with unresectable GIST. Tumor lysis syndrome in GIST has been not reported until 2000. Imatinib was first introduced in a patient with metastatic GIST in 2000, which demonstrated a rapid and sustained partial response [4]. Sunitinib was suggested to be used in imatinib-resistant GIST [5]. Owing to the potent effect, target molecular therapy is possible to cause tumor lysis syndrome in metastatic GIST. In 2007, Pinder, et al. [6] first reported the severe complication that an 81-year-old male patient with recurrent metastatic GIST, without liver metastasis, received imatinib 400 mg daily. Sunitinib 50 mg was also reported to cause fetal tumor lysis syndrome 7 days after initial treatment [7]. Our 43-year-old patient without systemic disease is the second metastatic GIST case, with liver metastasis and mild jaundice; treated with imatinib 400 mg daily had severe tumor lysis syndrome, renal failure and liver failure. Liver metastasis should be required special emphasis for the risk of tumor lysis syndrome in solid tumors [9].
The risk factors for tumor lysis syndrome in solid tumors are metastatic disease, a high tumor burden, and responsive to the applied anti-neoplastic treatments [9]. Our case was a metastatic GIST with high tumor burden, and effectively responsive to imatinib. These findings in our case are consistent with the above-mentioned risk factors for tumor lysis syndrome in solid tumors. Though, tumor lysis syndrome is rarely observed in solid tumors, however, the mortality rate in solid tumors complicated with tumor lysis syndrome is higher than that in hematological malignancies [9]. Reduced dose of imatinib 300 mg therapy and pre-treatment of aggressive hydration, rasburicase and allopurinol may be the optative prevention strategy to reduce mortality of tumor lysis syndrome in solid tumor after target molecular therapy [8]. Recognition of risk factors and appropriate interventions are the keys to prevent tumor lysis syndrome. According to the patient stratification by risk in evidence-based review study [10], solid tumors having the characteristics of rapidly proliferating with expected rapid response to therapy are categorized the intermediate risk for development of tumor lysis syndrome. Our case, featuring rapidly proliferating GIST with effective response to imatinib, is classified to the intermediate risk. For the patient with intermediate risk, hydration and allopurinol may be used for the prevention therapy of tumor lysis syndrome [10].
In conclusion, we reported a comparative young case of metastatic GIST and liver metastasis that develops tumor lysis syndrome and liver failure after treatment with imatinib 400 mg. Metastatic GIST is of intermediate risk of tumor necrosis syndrome. Reduced dose of imatinib 300 mg therapy and pre-treatment of aggressive hydration, rasburicase and allopurinol may be the prevention strategy.
Author Contributions
Yuan JN, and Chen CY were involved in the clinical care of patient; Li AFY analyzed pathological pictures; and Yuan JN and Chen CY wrote the paper.
References
- Mirrakhimov AE, Ali AM, Khan M, et al. (2014) Tumor lysis syndrome in solid tumors: an up to date review of the literature. Rare Tumors 6: 5389.
- Cairo MS, Bishop M (2004) Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol 127: 3-11.
- Davidson MB, Thakkar S, Hix JK, et al. (2004) Pathophysiology, clinical consequences, and treatment of tumor lysis syndrome. Am J Med 116: 546-554.
- Joensuu H, Roberts PJ, Sarlomo-Rikala M, et al. (2001) Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N Engl J Med 344: 1052-1056.
- Raut CP, Morgan JA, Ashley SW (2007) Current issues in gastrointestinal stromal tumors: incidence, molecular biology, and contemporary treatment of localized and advanced disease. Curr Opin Gastroenterol 23: 149-158.
- Pinder EM, Atwal GS, Ayantunde AA, et al. (2007) Tumour lysis syndrome occurring in a patient with metastatic gastrointestinal stromal tumour treated with Glivec (Imatinib Mesylate, Gleevec, STI571). Sarcoma 2007: 82012.
- Saylor PJ, Reid TR (2007) Tumor lysis syndrome after treatment of a gastrointestinal stromal tumor with the oral tyrosine kinase inhibitor sunitinib. J Clin Oncol 25: 3544-3546.
- Terada S, Matsuyama Y, Onoda K, et al. (2014) Successful re-administration of imatinib without recurrence of tumor lysis syndrome in a patient with peritoneal gastrointestinal stromal tumor. Nihon Shokakibyo Gakkai Zasshi 111: 1782-1788.
- Gemici C (2006) Tumour lysis syndrome in solid tumours. Clin Oncol (R Coll Radiol) 18: 773-780.
- Coiffier B, Altman A, Pui CH, et al. (2008) Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence-based review. J Clin Oncol 26: 2767-2778.
Corresponding Author
Chih-Yen Chen, MD, PhD, Division of Gastroenterology and Hepatology, Department of Medicine, Taipei Veterans General Hospital; Faculty of Medicine, National Yang-Ming University School of Medicine, 201, Sec. 2, Shih-Pai Road, Taipei 112, Taiwan, Tel: +886-2-28712121, Ext: 3763, Fax: +886-2-28711058.
Copyright
© 2017 Jeng-Nian Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.