Successful Treatment of Neonatal Extreme Breastfeeding-Associated Hypernatremia
Abstract
Exclusive breastfeeding is a leading cause of hypernatremia and dehydration. We describe successful treatment in a case of extreme breastfeeding hypernatremia in a neonate, presenting with serum sodium up to 232 mmol/L, acute renal failure and a rare complication of sinus vein thrombosis. With careful treatment, there were both clinical and laboratory improvement, with no further short-term complications proving that with attention, even extreme hypernatremia is treatable.
Keywords
Breastfeeding associated hypernatremia, Hypernatremia, Exclusive breastfeeding
Abbreviations
BFAH: Breastfeeding Associated Hypernatremia; PICU: Pediatric Intensive Care Unit
Introduction
Hypernatremia defined as serum sodium > 145 mmol/liter (neonatal hypernatremia is defined as serum sodium > 150) is one of the most common electrolyte disorders and is often due to net water loss. Since sodium is a functionally impermeable solute, high levels of extracellular sodium induce the movement of water across cell membranes resulting in cellular dehydration [1].
In the past, diarrheal dehydration was the most common cause for hypernatremia in infants [2]. Recent studies show that inadequate fluid intake and an iatrogenic cause are the leading causes for hypernatremia in infants today and is often accompanied by comorbidities [3]. Breastfeeding-associated hypernatremia (BFAH) is caused by insufficient breastfeeding due to maternal or infantile reasons and is aggravated when less milk is produced thereby resulting in higher sodium content [4]. Though different studies [4-6] place the incidence of BFAH at 0.1%-2.7% of term babies, some estimate that up to 10% of exclusively breastfeed babies suffer from BFAH [4]. During the last decades both exclusive breastfeeding and the incidence of BFAH has risen [4,7]. Morbidity is significant but mortality is very low when appropriate treatment is promptly administered [4,7].
Case Report
MN, an eight day-old male, presented at our emergency room with extreme hypernatremia and was admitted to the pediatric intensive care unit (PICU) for treatment and monitoring. The pregnancy was unremarkable and the patient was born during the 39th week of gestation following normal vaginal delivery, and a large for gestational age (LGA) birth weight of 4160 grams. He was the first child to young non-consanguineous parents and was exclusively breastfed at home.
During the three days prior to his admission, his parents reported poor appetite and weak sucking. On the day before presentation, he developed fever of up to 38.5 °C with cyanosis, but according to the family he had a fair amount of urine. There were no gastrointestinal symptoms. Additionally, the baby began grunting on the day of presentation and felt very cold to his parents. He was subsequently taken to an outpatient clinic of a local general hospital.
At first assessment, he was found to be tachypneic, cyanotic, stiff and irritable, with a prolonged capillary refill time of 6 seconds. His vital signs were a heart rate of 155 bpm, blood pressure was unmeasurable and his core temperature was 33.4 °C. He weighed 2900 g (a 30% decrease from birth weight). His skin was dry, doughy with a decreased turgor and his mucous membranes were extremely dry. The examination of his heart, lungs and abdomen was unremarkable.
Neurologically, his fontanelles were flat, he was irritable, with hyperflexion of his upper limbs, and though his Moro reflex was normal, he had a decreased sucking reflex.
Laboratory tests were performed including basic chemistry, complete blood count, arterial blood gases, and blood cultures. He was immediately started on normal saline fluid resuscitation, cefotaxime, ampicillin and oxygen, and was placed in an incubator for controlled elevation of body temperature. After fluid expansion with 40 cc/kg, his blood pressure could be measured (73/40 mmHg) and his core body temperature improved.
Initial laboratory results showed a sodium level of 200 mmol/liter, potassium of 5.4 mmol/liter, chloride of 152 mmol/liter, creatinine of 528 micromol/liter, BUN of 147 mg/dL and albumin 42 gram/L. He had a metabolic acidosis, with a pH of 7.17.
After calculation of water and sodium deficit, he was continued on normal saline at a rate of 38.5 cc/hr and was transferred to our hospital's PICU. The parents reported no use of milk formula, additional salt in the patient's diet, or any other additives.
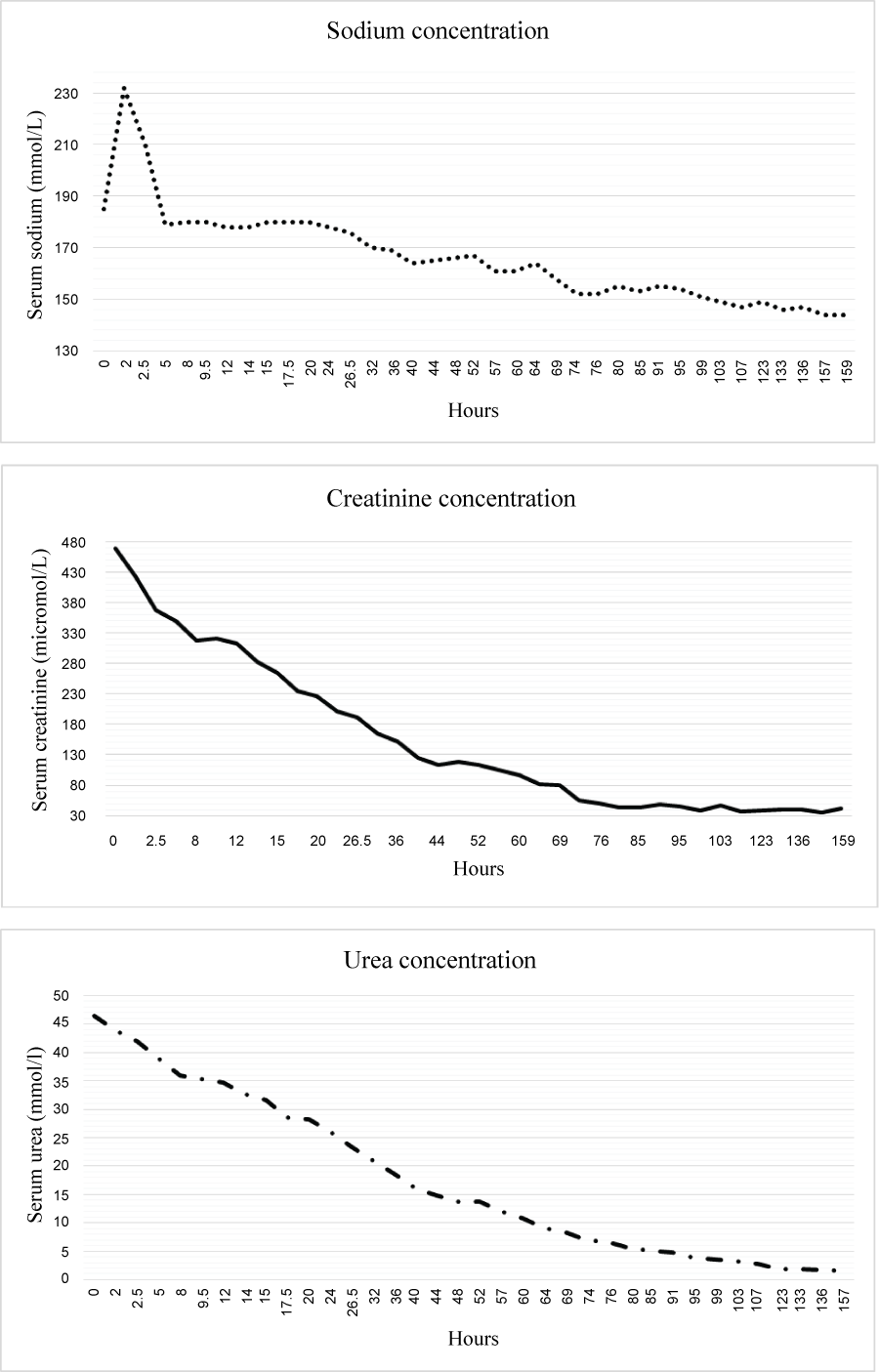
On presentation at the PICU, he was alert, with vital signs as follows: heart rate 130 bpm, blood pressure 70/40 mmHg, 36.5 °C core body temperature and O2 saturation in the 90 s, with a weight of 3100 g. He was treated according to the standard protocols for the treatment of hypernatremia. His water deficit was calculated around 700 cc and intravenous normal saline-half his deficit plus maintenance-was administered over 24 hours at a rate of 30 cc/hr. Electrolytes were sampled every two hours, and sodium concentration was normalized over the course of six days (Figure 1). The infant's kidney function returned to normal five days after presenting to our hospital. After correction of the severe dehydration, albumin levels dropped to 28 gram/liter, most likely because the first measured value was due to hemoconcentration, and did not accurately represent albumin levels. Several hours after admission, MN developed focal tonic seizures. His fontanelles were normotensive at this time and there were no changes in his primitive reflexes. He was treated with a loading dose of phenobarbital. Sodium at the time of the seizures was 180 mmol/L. Glucose and calcium levels were in the normal range. Brain computed tomography scan (without contrast) completed an hour after the seizures began, showed hyperdensity within the superior sagittal sinus extending from the frontal region through the posterior aspect of the vertex and associated with hyperdensities within the bilateral frontal vertex feeding veins suggesting a sinus vein thrombosis which was later confirmed by magnetic resonance imaging (MRI). No central pontine myelinolysis or brain edema was noted and was then started on a therapeutic dose of enoxaparin.
It should be noted that although the overall reduction in sodium concentration proceeded as expected, there were two abnormal sodium results, 230 mmol/L and 210 mmol/L at 2 and 2.5 hours respectively after admission to PICU. Diabetes insipidus and hormonal causes for hypernatremia were ruled out. It is important to note that as his mother's first-born child who was exclusively breastfed, MN was at risk for hypernatremia.
A month after first presenting to our hospital MN was discharged with no gross neurological deficit. The parents were counseled as to proper feeding for their child, and were guided to feed him with both breastfeeding and milk formula. At follow up six months after his first admission, he was, again, grossly neurologically intact.
Discussion
The advantages of breastfeeding are many and include reduced risk for infections, allergic diseases, diabetes, obesity, malignancy, sudden infant death and necrotizing enterocolitis. Further, there is lower overall mortality in breastfed infants as well as short- and long-term maternal benefits [8,9].
A 2012 Cochrane review [9] determined that the optimal duration of exclusive breastfeeding is six months. Breastfeeding-associated hypernatremia is caused by insufficient breastfeeding due to maternal or infantile reasons [4] though studies have shown an incidence of up to 2.7%-this is probably an underestimation. During the last decades both exclusive breastfeeding and the incidence of BFAH has risen, in all likelihood due to the public's response to the strong recommendations supporting exclusive breastfeeding for nearly all newborns [4,7,8].
Incidence is higher with primiparous mothers, uneducated mothers or when there are difficulties with breastfeeding, excessive room heating, oliguria, and inadequate stooling. Some studies show higher rates of BFAH following Caesarian section deliveries while others show higher rates of the illness after vaginal delivery [4-6,10]. The symptoms in infants are restlessness, lethargy, neurologic deterioration, seizures, intracranial hemorrhage, vascular thrombosis, coma and even death [2-4]. Weight loss > 10% was seen in all cases of BFAH [5,6,10]. In the case we report, the infant had many of the classic signs and symptoms.
Prognosis of hypernatremia depends on the underlying condition that led to the hypernatremia, its severity and the rapidity of onset [1]. Peak serum sodium does not correlate with mortality [4].
Resolving hypernatremia requires both treatment of the underlying cause as well as the correction of the high sodium serum levels and hypertonicity. For patients with acute onset of hypernatremia rapid correction is favored since chronic changes in brain fluid and electrolytes balance have not yet occurred. In patients with chronic or unknown duration of hypernatremia a maximal rate correction of 0.5 mmol/liter/hour and no more than 10 mmol/liter/day is recommended to avoid cerebral edema and convulsions caused by rapid rectification of the electrolyte abnormality. The correction should be done with the minimal amount of hypotonic fluids to reduce the risk for cerebral edema [1].
In our patient serum sodium was measured using indirect ion-selective electrodes (cobas® 6000 analyzer, Roche Diagnostics) and every abnormal result was rechecked twice or three times. However, this instrument's reliability is reported by the manufacturer to be confined to the 80 and 180 mmol/liter range only (Cobas® 6000 analyzer manual). Consequently, the results of its analysis had to be interpreted cautiously in MN's case as during his hospitalization his sodium levels changed dramatically with every test and reached an extreme value of 232 mmol/L. None of the studies we found reported sodium levels greater than 214 mmol/L [4-7,10].
In our view these high and fluctuating values were not solely attributable to the infant's clinical state or response to the treatment but at least in part to the unreliability of using indirect ionselective electrodes as a measurement tool. In evaluating the extremely high sodium values that were recorded we also had to consider the possible effect of mild hypoalbuminemia observed at presentation. This factor has been previously shown to increase indirect ion selective electrodes measurement of sodium [11].
Conclusions
We present a newborn infant, with normal prenatal and delivery history who suffered from extreme hypernatremia of up to 232 mmol/L and a rare complication of sinus vein thrombosis. He was diagnosed with BFAH due to his history of poor exclusive breast milk feeding and after ruling out alternative diagnoses. Despite the severity of his condition he gradually improved after undergoing judicious treatment according to established protocols. He was discharged from the hospital and on follow-up six months later displayed no neurological deficit.
This extreme case highlights several important points: (1) Pediatricians must be aware that BFAH is a common condition that requires appropriate parental guidance and close monitoring especially in high-risk cases; (2) Hypernatremia is a curable disorder when treated appropriately and expeditiously, and; (3) attention should be given to discrepancies in serum sodium ranges, especially concerning extreme values, when using ion-selective measuring instruments. The possibility of inaccurate measures reported by these instruments requires particular care and clinical evaluation on the part of the attending physician.
Conflict of Interest
All authors have indicated they have no potential conflicts of interest to disclose.
Funding
No external funding for this manuscript.
Financial Disclosure
All authors have indicated they have no financial relationships relevant to this article to disclose.
Contributors' Statement
Dr. Braun and Dr. Zilkha conceptualized and designed the study and reviewed and revised the manuscript.
Dr. Gross and Dr. Siedner-Weintraub collected data and drafted the initial manuscript.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Dr. Gross wrote the first draft of the manuscript. No honorarium, grant, or other form of payment was given to anyone to produce the manuscript.
References
- Adrogué HJ, Madias NE (2000) Hypernatremia. N Engl J Med 342: 1493-1499.
- Finberg L, Harrison HE (1955) Hypernatremia in infants an evaluation of the clinical and biochemical findings Accompanying this State. Pediatrics 16: 1-14.
- Moritz ML, Ayus JC (1999) The changing pattern of hypernatremia in hospitalized children. Pediatrics 104: 435-439.
- Moritz ML, Manoel MD, Bogen DL, et al. (2005) Breastfeeding-associated hypernatremia: are we missing the diagnosis? Pediatrics 116: e343-e347.
- Manganaro R, Mami C, Marrone T, et al. (2001) Incidence of dehydration and hypernatremia in exclusively breast-fed infants. J Pediatr 139: 673-675.
- Yaseen H, Salem M, Darwich M (2004) Clinical presentation of hypernatremic dehydration in exclusively breast-fed neonates. Indian J Pediatr 71: 1059-1062.
- Cooper WO, Atherton HD, Kahana M, et al. (1995) Increased incidence of severe breastfeeding malnutrition and hypernatremia in a metropolitan area. Pediatrics 96: 957-960.
- Section on Breastfeeding (2012) Breastfeeding and the use of human milk. Pediatrics 129: e827-e841.
- Kramer MS, Kakuma R (2012) Optimal duration of exclusive breastfeeding. Cochrane Database Syst Rev.
- Caglar MK, I Özer, Altugan FS (2006) Risk factors for excess weight loss and hypernatremia in exclusively breast-fed infants. Braz J Med Biol Res 39: 539-544.
- King RI, Mackay RJ, Florkowski CM, et al. (2013) Electrolytes in sick neonates–which sodium is the right answer? Arch Dis Child Fetal Neonatal Ed 98.
Corresponding Author
Itai Gross, Department of Pediatrics, Hadassah Medical Center, Ein Kerem, Kiryat Hadassah, POB 12000, Jerusalem, 91120, Israel, Tel: +972-50-5172-399, Fax: +972-2-533-4029.
Copyright
© 2017 Gross I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.