Unmasking Nephrotic Syndrome
Clinical Presentation
A 65-year-old male was referred for nephrology consult for increasing creatinine from 1.7 mg/dl to 4.4 mg/dl over a period of 4 months. The patient was not diabetic but had a history of hypertension and dyslipidemia for which he was treated with amlodipine and atorvastatin respectively. He had noted increasing body malaise, weight loss, poor appetite and bipedal edema. He also noted frothy urine. On physical examination his blood pressure was 140/90 mmHg. He had pale palpebral conjunctivae and bipedal edema. The rest of the physical exam was unremarkable. Laboratory examinations were done at the time of consultation (Table 1).
Renal Ultrasound: Normal sized kidneys
R Kidney 9.9 × 3.7 cm 1.6 cm cortex
L Kidney 9.7 × 4.7 cm 1.7 cm cortex
Discussion
1. What are the possible causes for nephrotic syndrome?
2. What are and are not the indications for percutaneous renal biopsy?
3. What is the role of serum free light chain assay?
4. What is the diagnosis?
What are the possible causes for nephrotic syndrome?
Nephrotic syndrome is characterized by five principal clinical or biochemical findings which include: High-grade, albumin dominant proteinuria (generally more than 3000 mg/day), hypoalbuminemia, edema, hyperdyslipidemia and lipiduria [1]. All of these are present in the patient. There are a large number of possible etiologies for nephrotic syndrome (Table 2) therefore, appropriate laboratory investigation together with a careful history and physical examination are important in determining its etiology.
Additional studies were performed to determine the cause of nephrotic syndrome in the patient (Table 3).
What are and are not indications for performing a percutaneous renal biopsy?
Renal biopsy is indicated in most adults with idiopathic nephrotic syndrome. Other indications include acute nephritic syndrome, unexplained renal impairment of a native or transplanted kidney, renal manifestation of systemic disease [2,3]. Renal biopsy may not be indicated in isolated microscopic hematuria, isolated non-nephrotic proteinuria (500-1000 mg/day), slowly progressive renal failure with a known etiology such as diabetes or hypertension or in acute renal failure with an obvious antecedent event such as hypotension [2,4]. Since the patient had nephrotic syndrome without an obvious cause he underwent a percutaneous renal biopsy with the following results:
Light microscopy: 8/9 glomeruli were globally sclerosed with mesangial matrix expansion, thickening of capillary loops by pale eosinophilic material. Tubular atrophy and tubular fibrosis (50%). Silver stain showed segmental spicule formation. Congo red stain focally positive for amyloid.
Immunoflourescence microscopy: Negative staining for IgG, IgA, Trace mesangial staining for IgM, C1q, C3, Fibrinogen, Cast staining + 3 for kappa and lambda light chains, albumin.
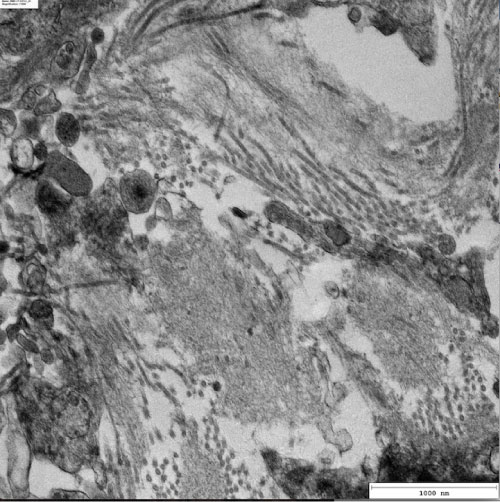
Electron microscopy: Non-recognizable foot processes. There are haphazardly arranged, rigid, non-branching fibrils measuring 9 nm in mean diameter (7-15 nm) in the glomerular basement membrane, mesangium, bowman's capsule, tubular basement membrane and interstitium (Figure 1).
What is the role of the Free Light Chain assay in the diagnosis and management of the patient?
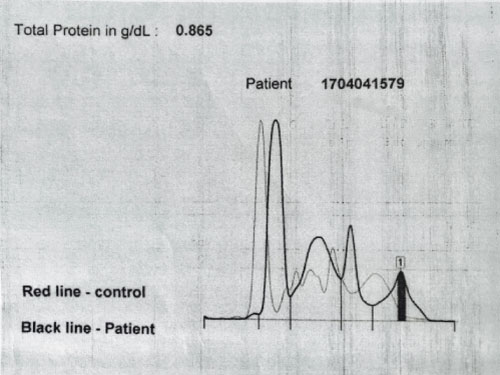
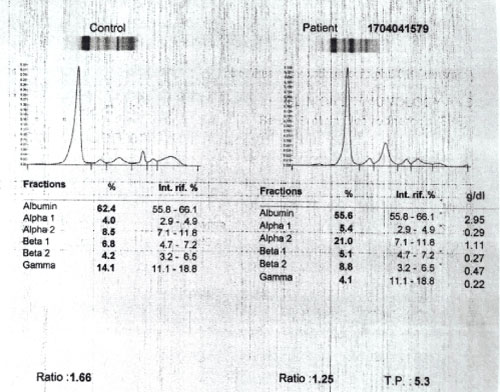
A plasma cell disorder was suspected thus the patient underwent a serum free light chain assay (Table 4), urine protein electrophoresis (Figure 2) and serum protein electrophoresis (Figure 3).
The serum free light chain analysis in combination with serum protein electrophoresis is a sensitive tool for the screening of most plasma cell disorders. The sensitivity of serum free light chain testing is generally greater than 90% in amyloid light chain (AL) amyloidosis where these assays often detect disease despite negative serum and/or urine immunofixation. Therefore, urine and serum studies are complementary tests when evaluating patients with suspected AL amyloidosis [5]. Free light chain testing is considered abnormal when the clonal free light chain is increased above normal and the free kappa/free lambda ratio is abnormal. A clonal free light chain concentration of at least 100 mg/L with an abnormal ratio is consistent with measurable disease [5].
A different set of reference values have been proposed for patients with chronic kidney disease because renal impairment may increase the concentrations of free kappa and free lambda light chains due to increased immune production or decreased clearance [5]. Thus using these reference values, the patient had a markedly elevated free lambda light chains however the free kappa/free lambda ratio was normal.
Another clinical utility for serum free light chain assays is to monitor response to chemotherapy. The International Myeloma Work Group (IMWG) advocates serial monitoring with serum FLC analysis for patients with oligosecretory plasma cell disorders including AL amyloidosis [5].
Serum FLC analysis can be also be used as a prognostic tool in AL amyloidosis. Prognosis is worse in patients with amyloidosis whose baseline concentrations of clonal free light chains are markedly increased or whose ratio is abnormal at baseline [5].
What is the diagnosis?
The patient has renal amyloidosis based on the clinical presentation of nephrotic syndrome, progressive renal dysfunction and histopathologic findings of nodular glomerulosclerosis with tubular fibrosis, Congo red positivity and the presence of rigid non-branching fibrils on electron microscopy. The free light chain assay with elevated free lambda light chains, monoclonal spikes in the UPEP and SPEP also support the diagnosis of renal amyloidosis.
References
- Emmett M, Fenves A, Schwartz J (2016) Brenner and rector's the kidney. (10th edn), Elsevier, Philadelphia, 760-762.
- Whittier W, Korbet S (2018) Indications for and complications of renal biopsy. UpToDate.
- Salama, A, Terence CH (2016) Brenner and rector's the kidney. (10th edn), Elsevier, Philadelphia, 915-924.
- Bandari J, Fuller T, Turner II R, et al. (2016) Renal biopsy for medical renal disease: Indications and contraindications. Can J Urol 23: 8121-8126.
- Seigel D, Bilotti E, van Hoeven K (2009) Serum free light chain analysis for diagnosis, monitoring, and prognosis of monoclonal gammopathies. Lab Medicine 40: 363-366.
Corresponding Author
Mayleen Jeniffer L Laico, M.D., F.P.C.P., F.P.S.N., Chong Hua Hospital, Cebu City, Philippines.
Copyright
© 2020 Laico MJL. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.