Thoracic Endometriosis in Monozigotic Twins
Abstract
Introduction
Endometriosis consists on the presence of functionally active ectopic endometrial tissue. The thorax is the most frequent extrapelvic location. The causes of endometriosis are not well known yet and involve both genetic and environmental factors. The aim of this article is not only to describe the case of thoracic endometriosis in two monozygotic twins, but also try to establish a relationship between: coincident characteristics and genetics, differences between both cases with environmental factors.
Case report
Case 1
A 34-year-old woman was referred to our service with intense dysmenorrhea and dyschezia. The examination revealed a 3 cm nodule in the recto-vaginal septum. Laparoscopy was performed, with excision of endometriotic implants in torus uterinus and uterosacral ligaments. In 2013 she suffered three episodes of spontaneous pneumothorax on the right side, treated by Thoracic Surgery using VATS. The anatomopathological study showed the presence of endometriotic implants in the diaphragm.
Case 2
Her twin sister, a 42-year-old woman was referred to our Unit. She had previous history of chest pain associated with menstruation and recurrent right pneumothorax in 2014, requiring surgery using VATS. She presented with intense pelvic pain and dyschezia. Laparoscopic surgery was performed, with hysterectomy, bilateral annexectomy and resection of endometriotic implants. The anatomopathological study confirmed the diagnosis of endometriosis.
Discussion
Endometriosis is a complex disease with a wide spectrum of clinical and anatomical manifestations. The etiology of endometriosis is not well known yet because it is multifactorial and would imply genetics and environmental factors. Several cases of endometriosis have been reported in monozygotic twins, estimating the risk in these cases between 75-87%. There are no previous descriptions of cases of twins with thoracic endometriosis. Thoracic endometriosis was first described in 1938 by Schwarz. Different genes and environmental factors may influence in the development of thoracic endometriosis in different ways; these facts could explain the wide variation in its expression. Due to the difficulty of its diagnosis, the identification of thoracic endometriosis is normally delayed until the temporal relationship of symptoms with the menstrual cycle is established. A multidisciplinary treatment is neccesary. This treatment involves participation of several medical services, and a medical and surgical approach.
Introduction
Endometriosis consists on the presence of functionally active ectopic endometrial tissue. It usually affects the pelvic organs for example: ovary, anterior and posterior cul-de-sac, posterior broad ligaments and uterosacral ligaments [1]. Within the extrapelvic location, the thorax is the most frequent one, and it may compromise the diaphragm, pleura, pulmonary parenchyma and bronchi [2].
The causes of endometriosis are not well known yet. It is a complex entity whose etiology could involve both genetic and environmental factors, both of them contributing in an equivalent way to the development of the disease [3]. Several studies have been conducted in order to know its genetics. In studies performed in monozygotic twins, a high concordance between both twins for the appearance of endometriosis was found but, to a lesser extent, for their severity [4,5]. Additionally, in these studies, the occurrence of thoracic endometriosis in monozygotic twins is not mentioned.
The aim of this article is firstly to describe the case of thoracic endometriosis in two monozygotic twins, the symptoms and the management that was taken in each of them. And eventually, try to establish a relationship between: coincident characteristics and genetics, the differences between both cases with environmental factors.
Case Report
We present the case of two monozygotic twins followed in our Endometriosis Unit, developing both thoracic endometriosis.
Case 1
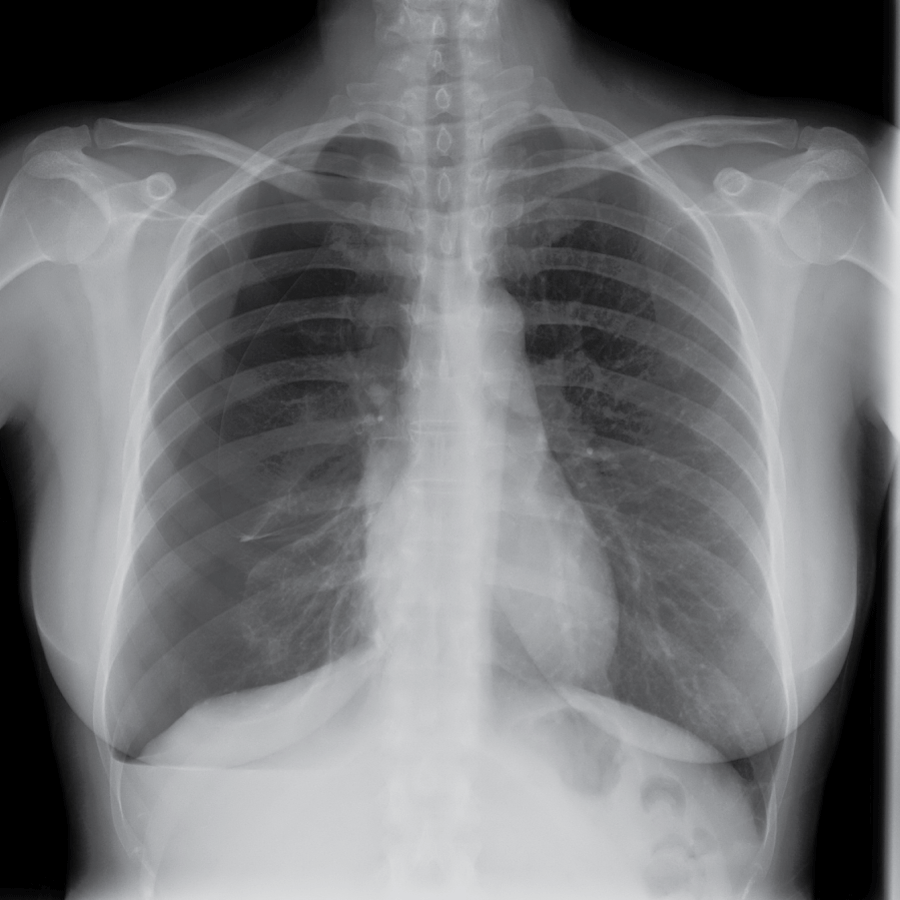
A 34-year-old woman, with a history of cesarean section, curettage due to miscarriage and laparoscopic cystectomy due to 4 cm endometrioma in 2006. She is referred to our service in 2007 with intense dysmenorrhea and dyschezia. The patient had been under treatment with oral contraceptives which she had left to try pregnancy. Gynecological examination and ultrasonography revealed a 3 cm nodule in uterosacral ligament involving rectum. Because of the symptoms of the patient and the suspicion of intestinal involvement, surgery was chosen. During laparoscopy, sigma adhesions were observed in the left iliac fossa and endometriotic implants in torus uterinus, uterosacral ligaments and rectal anterior wall. No lesions were found in diaphragmatic domes. Laparoscopy approach was done with adhesiolysis; dissection of both pararectal fossa. Ureters and hypogastric nerves were identified and dissected. Excision of endometriotic implants by skinning of rectal anterior wall was done. After the surgery, she received contraceptive treatment with total remission of pain. Afterwards, she left the treatment to try spontaneous gestation, that was achieved in 2010 and the delivery was by caesarean section. She remained asymptomatic, with no need for contraceptive treatment. Between March and August 2013, she suffered three episodes of spontaneous pneumothorax on the right side (Figure 1). She received treatment by Thoracic Surgery using Video-Assisted Thoracoscopic Surgery (VATS) with wedge resection in the upper lobe of the right lung, total pleural abrasion and diaphragmatic fenestration suture. The anatomopathological study showed the presence of endometriotic implants in the diaphragm. Medical treatment with GnRH analogues and hormonal replacement therapy with Tibolone was provided, and the patient appears asymptomatic in the follow-up from 2013 to the present.
Case 2
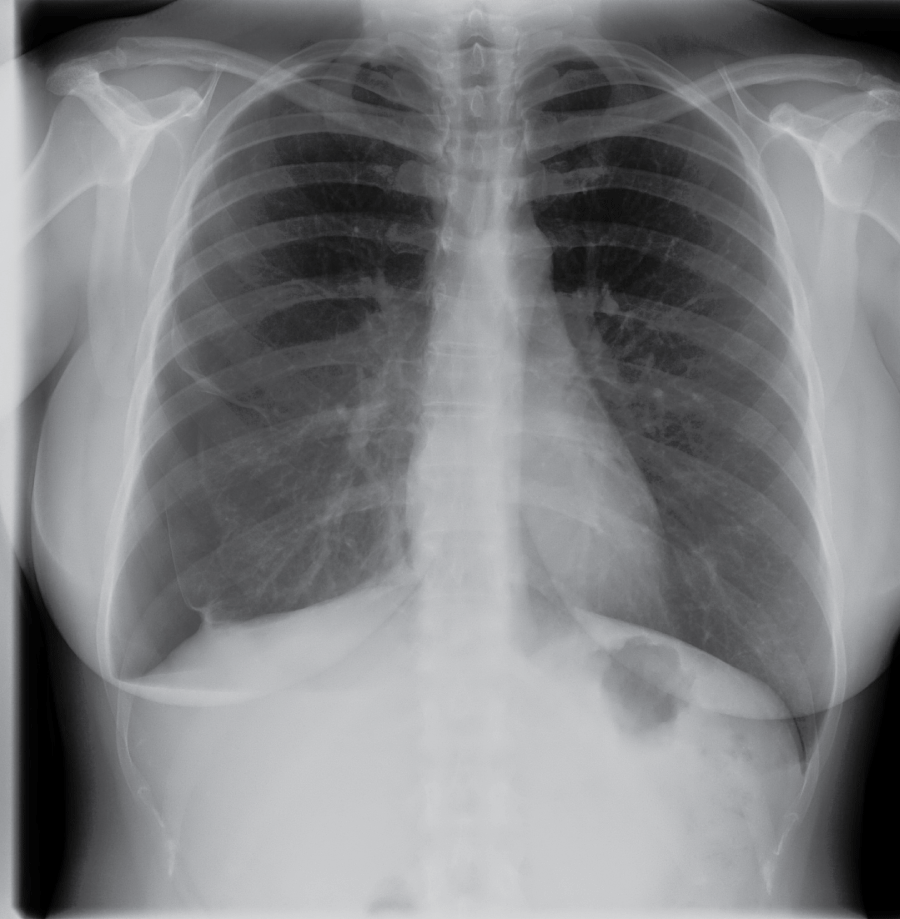
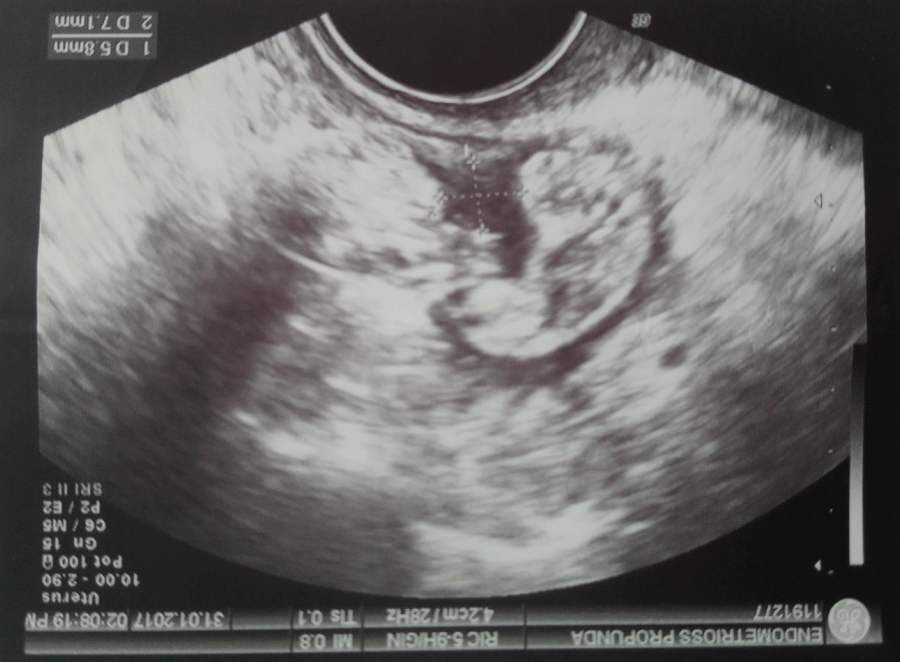
In 2015, her twin sister was referred to our Unit, a 42-year-old woman, with a history of cesarean section and posterior colporrhaphy in another center. She had a previous history of chest pain associated with menstruation and recurrent right pneumothorax in 2014 (Figure 2), requiring surgery using VATS, performing segmentectomy of right lower lobe and chemical pleurodesis with talc. The anatomopathological study described lymphocytic infiltration and non-specific fibrosis. The patient had been previously treated with oral contraceptives and therapy with GnRH analogues and Tibolone. She presented intense pelvic pain and dyschezia. The gynecological and ultrasound examination showed intense contraction of the muscles of the pelvic floor, vulvodynia, right hydrosalpinx and a 3-cm node in torus uterinus, which retracts intestine and compromises the bowel's muscle, without involvement of vaginal mucosa (Figure 3). Because of the inefficacy of the medical treatment and no more gestation desires, surgery was decided. Laparoscopic surgery was performed, and two endometriotic nodes were observed, one of 2 cm at the left posterolateral cervix, which strongly pulls the vagina and another on the anterior wall of the rectosigma, with intestine stenosis. We performed hysterectomy, bilateral annexectomy and resection of endometriotic implants, with rectosigma resection with end-to-end anastomosis. Vestibulectomy was also performed. The anatomopathological study confirmed the diagnosis of endometriosis. Three months after surgery, there is a significant improvement of the pain. She remains on treatment with estrogen-progestogen transdermally.
Discussion and Conclusion
Endometriosis is a complex disease with a wide spectrum of clinical and anatomical manifestations, prevailing in around 10% in women of reproductive age, with a mean age at diagnosis between 24-29 years [6].
The etiology of endometriosis remains still poorly understood, because it is multifactorial and could imply a genetic basis on which environmental factors act. Several endometriosis studies have been published in monozygotic twins [3-5,7], estimating the risk in these cases, between 75-87% [7]; much higher than in non-twin sisters. However, in these cases, the severity and location of endometriosis may differ in each twin. As far as we know, there are no previous descriptions of cases of twins with intestinal and thoracic endometriosis simultaneously.
Although genetic predisposition has been described and confirmed [8], it has not an immediate role in the prediction of the risk of endometriosis and, even less, for the prediction of special entities, as in this case, thoracic endometriosis.
Thoracic endometriosis was first described in 1938 by Schwarz [9], finding endometriosis in the lung parenchyma and later, in 1958, Maurer described the spontaneous pneumothorax associated with the menstrual cycle [10,11].
Although thoracic endometriosis is a rare clinical entity, it is the most common extra-pelvic location. It can affect the diaphragm, pleura, lung parenchyma and/or bronchi; and it may be present as catamenial pneumothorax, hemothorax, hemoptysis or the appearance of pulmonary nodes [2,12], affecting the right side in more than 80% of cases, as in our cases. It appears in young women, with an average age of 35 years (15-54 years), having a peak incidence about 5 years later than pelvic endometriosis [12]. In our case, the appearance of symptoms related to thoracic endometriosis was much later, 40 and 41 years, respectively. This fact can be explained because the onset of symptoms may go unnoticed if they are mild symptoms or because the relationship with menstruation is not established until later.
At the moment, the etiopathogenesis of this entity is not well known, postulating several theories [13,14]. Firstly, the Sampson theory, about auto transplantation by retrograde menses, through defects of the diaphragm, and to a greater extent on the right side due to the predisposition of the peritoneal fluid by the right pariethole gutter [11]. However, there are cases of thoracic endometriosis without defects of the diaphragm. Secondly, the microembolization of endometrial tissue through the lymphatic and/or venous system, mainly after a surgical procedure [6]. Finally, Ivanoff's theory of coelomic metaplasia, in which endometrial tissue could pass through diaphragmatic fenestrations and produce metaplasia of pleural surface [6].
There are known risk factors for thoracic endometriosis including infertility, prior gynecological surgery, and pelvic endometriosis [15,16]. In our cases, both patients had a history of cesarean section and the first one also had a cystectomy for endometrioma.
Therefore, different genes and environmental factors may influence the development of thoracic endometriosis in different ways, which might explain the wide variation in the expression of this syndrome. In the case exposed, both sisters had a shared genetic, which made them susceptible to present the disease, even the thoracic disease, but the different environmental factors or circumstances to which they were exposed could explain the differences they present. The main differences are the age of onset of the symptoms; and the presentation in the first case of pelvic endometriosis and seven years later the thoracic one, compared to the beginning of the thoracic symptoms prior to the symptoms of pelvic endometriosis in the second twin. This can also be justified by the fact that it is not possible to determine when the lesions appeared, and furthermore we only have information about when the symptoms involved a medical consultation.
The diagnosis of thoracic endometriosis is often difficult and is normally delayed until the temporal relationship of symptoms with the menstrual cycle is established. Definitive diagnosis requires histological confirmation, characterized by the classic triad of endometrial glands, stroma and macrophages; however, these three components do not always appear simultaneously [16,17]. We can talk about "thoracic endometriosis syndrome" when thoracic manifestations appear in association with the menstrual cycle, although we do not have such histological confirmation. In our first case, the anatomopathological study confirmed the diagnosis, but not in the second, where a nonspecific result was obtained.
Thoracic endometriosis needs a multidisciplinary approach, which involves the participation of several medical services, such as Gynecology, Thoracic Surgery and General Surgery [11]. The treatment of endometriosis begins with hormonal treatment, being the first line of treatment the combined contraceptives or progestin-only therapy. When these fail, more aggressive therapies are used, such as GnRH agonists, always in add-back therapy [18]. In cases where the pain is not controlled with medical therapy, surgery is used as a complementary treatment, like the cases described above. Surgery looks for, when possible, the complete excision of the pelvic and peritoneal implants by laparoscopy. In order to achieve this goal, large resections are needed sometimes, such as the recto-sigma resection performed in our second case. For thoracic endometriosis, the benefit of videothoracoscopy and chemical pleurodesis has been described for resection of endometriotic lesions and treatment and prevention of pneumothorax, respectively [11,19,20]. In the first case, given the age of the patient and the increase in cardiovascular risk of oophorectomy in women under 65 years, we chose to preserve ovaries and establish therapy with GnRH analogues and Tibolone. In the second case, due to the intense chronic pelvic pain and the complexity of the lesions, a more radical surgery was performed, and subsequent hormonal replacement therapy was established without any adverse effects at the moment.
References
-
Jansen RP, Russell P (1986) Nonpigmented endometriosis: clinical, laparoscopic, and pathologic definition. Am J Obstet Gynecol 155: 1154-1159.
-
Alifano M, Trisolini R, Cancellieri A, et al. (2006) Thoracic endometriosis: current knowledge. Ann Thorac Surg 81: 761-769.
-
Treloar SA, O'Connor DT, O'Connor VM, et al. (1999) Genetic influences on endometriosis in an Australian twin sample. Fertil Steril 71: 701-710.
-
Hadfield RM, Mardon HJ, Barlow DH, et al. (1997) Endometriosis in monozygotic twins. Fertil Steril 68: 941-942.
-
Saha R, Petterson HJ, Svedberg P, et al. (2015) Heritability of endometriosis. Fertil Steril 104: 947-952.
-
Agarwal N, Subramanian A (2010) Endometriosis-morphology, clinical presentations and molecular pathology. J Lab Physician 2: 1-9.
-
Moen MH (1994) Endometriosis in monozygotic twins. Acta Obstet Gynecol Scand 73: 59-62.
-
Rahmioglu N, Nyholt DR, Morris AP, et al. (2014) Genetic variants underlying risk of endometriosis: insights from meta-analysis of eight genome-wide association and replication datasets. Hum Reprod Update 20: 702-716.
-
Schwarz O (1938) Endometriosis of the lung. Am J Obstet Gynecol 36: 887-889.
-
Maurer ER, Schaal JA, Mendez FL Jr (1958) Chronic recurring spontaneous pneumothorax due to endometriosis of the diaphragm. J Am Med Assoc 168: 2013-2014.
-
Nezhat C, Main J, Paka C, et al. (2014) Multidisciplinary Treatment for Thoracic and Abdominopelvic Endometriosis. JSLS 18: e2014.00312.
-
Joseph J, Sahn SA (1996) Thoracic endometriosis syndrome: new observations from an analysis of 110 cases. Am J Med 100: 164-170.
-
Vinatier D, Orazi G, Cosson M, et al. (2001) Theories of endometriosis. Eur J Obstet Gynecol Reprod Biol 96: 21-34.
-
Ziedalski TM, Sankaranarayanan V, Chitkara RK (2004) Thoracic endometriosis: a case report and a literature review. J Thorac Cardiovasc Surg 127: 1513-1514.
-
Jubanyik K, Comite F (1997) Extrapelvic endometriosis. Obstet Gynecol Clin North Am 24: 411-440.
-
Ghigna MR, Mercier O, Mussot S, et al. (2015) Thoracic endometriosis: clinicopathologic updates and issues about 18 cases from a tertiary referring center. Ann Diagn Pathol 19: 320-325.
-
Flieder DB, Moran CA, Travis WD, et al. (1998) Pleuropulmonary endometriosis and pulmonary ectopic deciduosis: a clinicopathologic and immunohistochemical study of 10 cases with emphasis on diagnostic pitfalls. Hum Pathol 29: 1495-1503.
-
Johnson NP, Hummelshoj L, World Endometriosis Society Montpellier Consortium (2013) Consensus on current management of endometriosis. Hum Reprod 28: 1552-1568.
-
Nezhat C, Nicoll LM, Bhagan L, et al. (2009) Endometriosis of the diaphragm: four cases treated with a combination of laparoscopy and thoracoscopy. J Minim Invasive Gynecol 16: 573-580.
-
Inmaculada Duyos, Ana López-Carrasco, Alicia Hernández, et al. (2014) Management of thoracic endometriosis: single institution experience. Eur J Obstet Gynecol Reprod Biol 178: 56-59.
Corresponding Author
Silvia Marín Camacho, Obstetrics and Gynecology, University Hospital La Paz, Paseo de la Castellana, 261, Madrid, Spain, Tel: +34-660245064.
Copyright
© 2017 Marín CS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.