Simplified Surgical Technique for the Management of Severe Mitral Annulus Calcification
Abstract
Purpose
Severe mitral annular calcification (MAC) is occasionally associated with degenerative mitral valve dysfunction especially in the elderly and renal patients. Dealing with the MAC at the time of mitral valve replacement is difficult and hazardous.
Currently two techniques exist. The first involves extensive decalcification of the annulus followed by pericardial patching of the resulting gusset. The second technique is where the prosthesis is sutured within the atrium creating a new annulus. Complications of the first include atrio-ventricular disruption, circumflex artery injury and ventricular rupture while the second technique is associated with paravalvular leak, valve dehiscence and thromboembolism.
Methodology
We describe an extremely simple yet effective technique for valve replacement in the setting of severe MAC. The technique involves inserting the valve at its usual level within the annulus and native mitral valve with a pericardial collar that covers the severe MAC and is sutured to the atrium several centimetres from the prosthesis-sewing ring.
Results
Nine elderly patients underwent this procedure. All patients had surgery for mitral regurgitation or mixed mitral disease. There was one early post-operative death due to pulmonary artery rupture secondary to Swan-Ganz catheter injury. There were no other complications in the remaining eight, in particular no strokes. There were no paravalvular leaks seen on intra-operative TOE or early and late follow-up TTE. On long-term follow-up there has been no reoperations or complications associated with this technique.
Conclusion
This technique is simpler, safer and is associated with better outcomes than previous described techniques.
Keywords
Mitral valve, Mitral valve replacement, Annular calcification
Abbreviations
MAC: Mitral Annular Calcification; AV: Atrioventricular; TOE: Transoesophageal Echocardiogram; TTE: Transthoracic Echocardiogram; MR: Mitral Regurgitation; MVR: Mitral Valve Replacement
Introduction
Severe mitral annular calcification (MAC) is occasionally associated with degenerative mitral valve dysfunction especially in elderly women and renal patients [1]. MAC at the time of mitral valve replacement is difficult and hazardous.
The pathophysiology related to MAC is poorly understood and is most likely thought to be related to age related degeneration and reorganisation of collagen fibres in valve tissue [1]. MAC is usually a disease of later life and seen more predominately in females. MAC is not only associated with mitral valve dysfunction but also cardiovascular disease including coronary artery disease and stroke, arrhythmias and increased risks of complications associated with mitral valve repair/replacement [1-3]. The presence of MAC in patients when undergoing a mitral valve repair or replacement ranges from 19%-24% [3].
Currently there are two main techniques for dealing with severe MAC at the time of mitral valve replacement. The first involves extensive decalcification of the annulus and removing all of the calcium followed by a wide pericardial patch of the resulting gusset. This is difficult, time consuming and may be associated with AV disruption and thrombo-embolic events [2-4]. The second technique is where the prosthesis is not sutured at the level of the annulus but within the atrium, creating a new annulus. This has been associated with paravalvular leak, valve dehiscence and thromboembolism [3].
We described herein a simplified surgical approach to the patient with severe MAC.
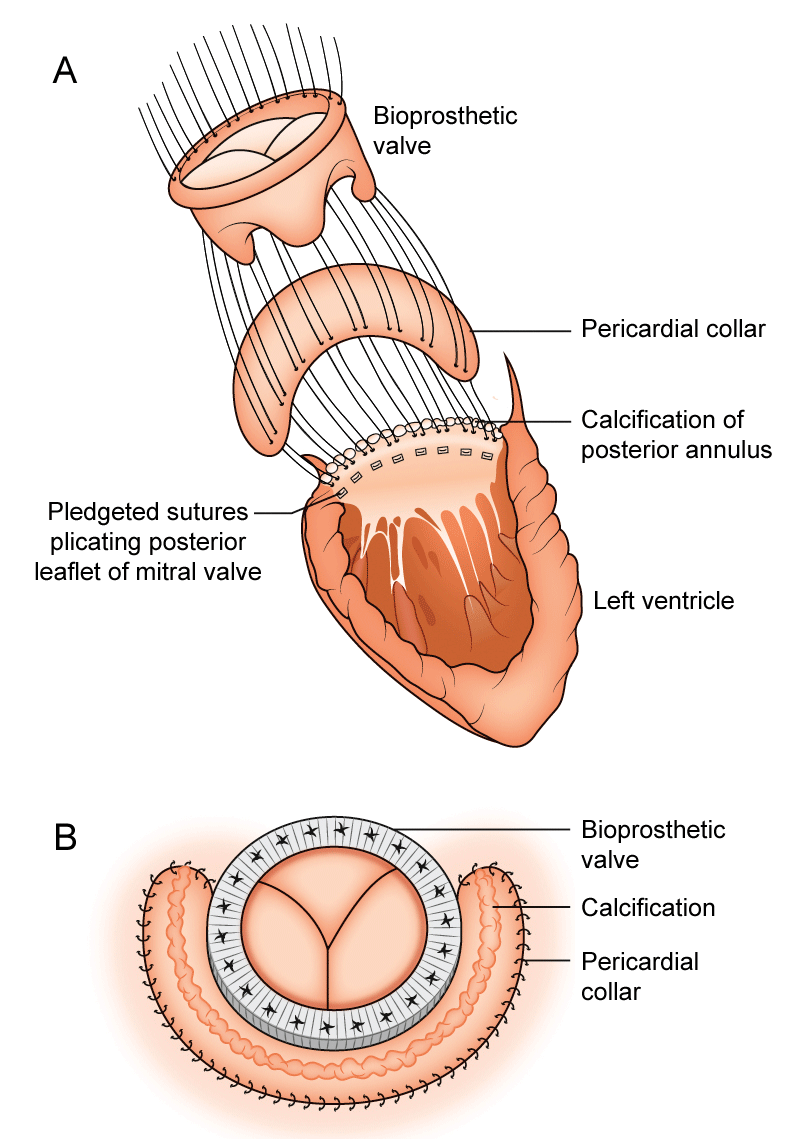
Surgical Technique
Calcification usually involves the posterior annulus to varying degrees but occasionally from trigone to trigone. The annular calcification extends into the posterior leaflet but the body and the free margin of the posterior leaflet are usually uninvolved. The entire anterior leaflet and associated subchordal structures are excised. 2.0 Ethibond pledgeted sutures are inserted in the anterior annulus in the usual fashion with the pledgets on the ventricular side. Multiple 2.0 Ethibond pledgeted sutures are then inserted into the posterior leaflet only, plicating the leaflets toward the annular calcification (Figure 1A). Again the plegets are on the ventricular side and the sutures do not enter the calcification, as this is likely to fracture the calcium and lead to thrombo-embolism. The calcium is left alone and intact. Often small tears in the posterior leaflet are noted but the majority of these sutures plicating the posterior leaflet are firm and this entire area will be reinforced by the pericardial collar.
We have used the Edwards pericardial Perimount tissue valve in all our cases and the valve size is selected after the sutures have been inserted. Bovine pericardial patch is then prepared and cut to size using the valve sizer to create a collar around the bioprosthesis. The collar usually extends from trigone to trigone but can be cut shorter or longer depending on the degree of calcification. The width of the collar is usually 2.5 cm-3.0 cm. The already placed valve sutures are inserted into the tissue bioprosthesis anteriorly as per usual. Posteriorly from trigone to trigone the sutures are inserted into the pericardial collar first and then into the sewing ring of the bioprosthesis (Figure 1A). The bioprosthesis with the pericardial collar are then seated so that the valve sits at the usual place, level of the mitral annulus. Posteriorly the sewing ring sandwiches the pericardial collar against the plicated leaflet with the edge of the sewing ring against the calcium (Figure 1B). The free edge of the pericardial collar is lifted to ensure that the sewing ring is down at the level of the annulus. The sutures are then tied as usual. A 4.0 prolene double ended suture is then inserted at each trigone from the atrium into the pericardial collar and then into the sewing ring and tied. A running continuous 4.0 prolene suture then securesthe free edge of the pericardial collar to the left atriumapproximately 2 cm from the sewing ring from trigone to trigone. A second layer of 4.0 prolene is performed to smooth out the edge of the pericardial patch.
Results
Nine elderly patients, one on haemodialysis, underwent this procedure over the last twelve years. Eight patients were female with a mean age of 79 ± 5 years. All patients underwent surgery for severe mitral regurgitation with severe mitral annular calcification. Concomitant surgery included cryoablation in 7, bypass surgery in 2, tricuspid annuloplasty in 1 and aortic valve replacement (AVR) in 1 See (Table 1). All patients with severe MR and severe MAC were included consecutively. Patients were excluded if the MAC had to be removed because an adequately sized valve could not be inserted, or there was endocarditis with MAC involvement.
There was one early post-operative death due to pulmonary artery rupture caused by a Swan-Ganz catheter. No other complications were evident in the remaining 8 patients, in particular no strokes were recorded. There have been no paravalvular leaks seen on intraoperative TOE or early and late follow up TTE. On long-term follow up (mean follow up 54 ± 56 months) there have been no complications associated with this technique or mitral valve reoperations.
Discussion
Current techniques for the management of severe MAC are complicated and associated with atrio-ventricular (AV) disruption, ventricular rupture, circumflex artery injury and higher rate of thrombo-embolic events. Our described technique is safe, simple and a reproducible [4].
With our technique the mitral valve prosthesis is seated at the annular level so that the struts are not at risk of being impinged by the leaflets or the calcium, which can occur with techniques where the valve is tilted or sutured within the wall of the atrium. There is also no significant dead space that leads to clot formation and thromboembolism. The pericardial collar is attached to the sewing ring via the plicated valve sutures and the collar is used to cover these sutures in case these sutures tear and cause paravalvular regurgitation. The pericardial collar also covers the calcified mitral annulus whilst the lateral margin is sutured to the left atrium approximately 2 cm from the annulus. Any small leaks after coming off bypass at the level of the leaflets will leak into a small dead space under the patch in the left atrium that would be of no consequence. We have not noted any intermediate or long-term issues with this pericardial collar.
Mihaljevic, et al. in 2013 described the complete debridement of the MAC before covering the resulting defect in the atrio-ventricular groove with a pericardial patch potentially resulting in thrombo-embolic events, AV disruption or circumflex artery injury [2]. Given that the calcification remains untouched in our approach we hypothesise that our rates of thromboembolic events will be lower than other techniques.
Our technique is extremely simple, safe and reproducible. The technique is usually possible, especially in patients with mitral regurgitation, where an adequately sized tissue valve can be inserted even when leaving the calcium in situ. Unfortunately this technique is occasionally not possible in some patients with severe mitral stenosis where even after the anterior leaflet has been excised an adequately sized tissue valve cannot be inserted and in these cases, the calcium needs to be removed. Also in patients with endocarditis, infection often involves the posterior leaflet and the mitral annular calcification and in these cases again, the calcium needs to be removed.
This procedure is extremely straightforward with no risk of AV disruption, ventricular rupture, circumflex injury, and an extremely low thromboembolic risk as the calcium has been left intact. We would recommend this technique in the majority of patients where an adequately sized valve can be inserted and those who do not have endocarditis as the safest technique for valve replacement in patients with severe MAC.
Acknowledgements
None.
References
- Carpentier A, Pellerin M, Fuzellier JF, et al. (1996) Extensive calcification of the mitral valve annulus: pathology and surgical management. J Thorac Cardiovasc Surg 111: 718-729.
- Mihaljevic T, Koprivanac M, Kelava M, et al. (2013) Mitral valve replacement in patients with severely calcified mitral valve annulus: Surgical technique. J Thorac Cardiovasc Surg 146: 233-235.
- Abramowitz Y, Jilaihawi H, Chakravarty T, et al. (2015) Mitral annulus calcification. J Am Coll Cardiol 66: 1934-1941.
- Uchimuro T, Fukui T, Shimizu A, et al. (2016) Mitral valve surgery in patients with severe mitral annular calcification. Ann Thorac Surg 101: 889-895.
Corresponding Author
Manu N Mathur, Department of Cardiothoracic Surgery, North Shore Private Hospital, St Leonards, NSW 2065, Suite 3, Level 5, Australia, Tel: +61422-306-162.
Copyright
© 2017 Bowers PJT, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.