Effects of Breast Radiotherapy on the Heart Ejection Fraction
Abstract
Background: Globally, breast cancer (BC) is the most common malignancy affecting women and is the foremost contributor to cancer-related deaths among the female population in both developed and developing regions. In Iraq BC constitute 18.96% of all other types of cancers up to 2011.
Aim of study : To shade the light on the radiation therapy effect on the ejection fraction of the heart in BC patients.
Methods : This is a prospective observational study that was conducted in Radiotherapy and nuclear medicine center of the oncology teaching hospital/medical city complex using Echo cardiograph for following up the patients. Data recruitment included patients received Adjuvant 3D conformal radiotherapy as a whole breast irradiation or as a chest wall irradiation from August/2016 to January/2017 and followed with Echo study four months later until January/2017.
Results : In our registry 50 patients were recorded; patient’s age was between 32 and 70 years old female gender. We found that there was a significant effect of radiation on the ejection fraction percentage of pre radiotherapy and post radiotherapy of both radiotherapy doses (P=0.001). We noticed that patients with left sided breast whom received 4260 cG had the highest significant result of a mean difference of pre-radiation =66%, and post radiotherapy =65.5%. Also we noticed that patients whom had received 5000 cG of either left or right sided BC, have substantial difference between pre-radiotherapy of 66.3% and 66.8% to 64.3% and 65.2% for right and left side respectively.
Conclusion : In patients with BC treated with adjuvant 3D conformal radiotherapy as a whole breast irradiation or as a chest wall irradiation, an evaluation for the cardiac ejection fraction after four months of completing their radiotherapy, to either right sided or left sided breast, there was a marked difference in the ejection fraction size between the left sided treated BC and right sided BC, and between different doses mainly due to scattered and transmitted irradiation to the coronary arteries.
Keywords
BC, RT, 3DCRT, EF, cardiac toxicity
Introduction
Each year, breast cancer is diagnosed in over one million women worldwide [1]. In Iraq BC accounts for 18.96% of all types of cancer registered in 2011 and it is the most common cancer between females [2]. In the early stage BC, BCS followed by RT is equivalent to mastectomy; also RT improves local control and survival for node positive women after mastectomy or BCS [1].
In locally advanced disease, usually the management starts with chemotherapy or hormonal therapy as systemic therapy, and then mastectomy and loco regional RT should follow systemic therapy [1]. There are studies conducted by many institutions around the world to show the effect of RT on the heart in BC patients, of those one had been conducted by Oxford University in 2013 that shows Exposure of the heart to ionizing radiation during RT for BC increases the subsequent rate of ischemic heart disease. The risk increases in proportion to the average radiation dose received by the heart, typically emerging within a few years post-exposure and persisting for at least two decades. Women with existing cardiovascular risk factors experience a higher absolute increase in risk from radiotherapy compared to those without such conditions [3]. Another recently published study by mayo clinic shows the relative risk of heart failure and pathological ejection fraction increases with increasing cardiac radiation exposure during contemporary conformal BC RT [4].
It was traditionally thought that any loss of myocytes would be offset by fibrosis or hypertrophy of remaining myocytes. However, this perspective-based on the assumption that the heart is a post-mitotic organ-has been challenged by studies identifying cardiac stem cells capable of regeneration in the adult heart [5]. Cells critical to cardiac function, such as endothelial and connective tissue cells, exhibit very limited proliferative capacity. Based on the theory that radiation primarily affects rapidly dividing tissues, the heart was long believed to be relatively resistant to therapeutic levels of radiation. However, beginning in the late 1960s, experimental work by Fajardo and Stewart provided evidence that both the heart and its vasculature are indeed radiosensitive structures [6]. Pericarditis was initially recognized as the most frequent clinical manifestation, particularly when large portions of the heart-specifically the pericardium-were exposed to radiation doses exceeding 40 Gy [7]. Early research identified only a small number of coronary artery disease cases directly linked to radiation exposure. However, it is now understood that radiation-induced heart disease (RIHD) encompasses a broad range of cardiac complications. These include pericarditis, cardiomyopathy, myocardial and coronary artery disease, pericardial effusion or constrictive pericarditis, valvular dysfunction, and various arrhythmias [8]. Although most extensively investigated in patients with Hodgkin lymphoma and breast cancer, this phenomenon can arise following radiation therapy for any thoracic malignancy, including lung cancer, esophageal carcinoma, and thymoma [9]. Radiation can affect all anatomical components of the heart. In the pericardium, it may lead to pericarditis accompanied by effusion, with or without progression to constrictive pericarditis [10]. Involvement of the myocardium can result in cardiomyopathy, often driven by radiation-induced macroangiopathy and fibrosis, which may ultimately lead to heart failure. Vascular damage includes radiation-induced acceleration of atherosclerosis, increasing the risk of coronary artery disease and carotid artery disease, and thereby raising the likelihood of ischemic heart disease (IHD) and ischemic stroke, respectively [11].
Radiation has been identified as an independent risk factor in the development of atherosclerosis, acting synergistically with established cardiovascular risk factors [12]. One hypothesis proposes that atherosclerosis is a monoclonal process-similar to cancer-originating from a single mutated cell triggered by an external factor such as radiation. This may help explain the elevated risk of cardiovascular events even following exposure to low doses of radiation [13]. However, studies on human smooth muscle cells suggest they are monoclonal by nature, potentially indicating clonal expansion of pre-existing cells rather than radiation-induced mutations [14]. Genomic instability, observed in both radiation exposure and atherosclerosis, may represent a shared underlying mechanism linking the two conditions [15]. Early randomized trials of radiotherapy (RT), along with meta-analyses by the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), demonstrated reductions in breast cancer mortality, though these were offset by increased cardiovascular deaths [16]. Notably, patients with left-sided breast cancer have been shown to experience a higher incidence of ischemic heart disease (IHD)-related mortality compared to those with right-sided disease [17-18].
Regional perfusion defects corresponding to the radiation fields in the anterior part of the left ventricle of the heart were found and this was considered as an indicator of microangiopathy. The perfusion defects may persist or may appear 3-6 years post RT. The clinical significance of this finding is unclear, since the perfusion defects were not associated with changes in regional wall motion or EF [20-22].
In the context of RT optimization, 3DCRT frequently achieves adequate target volume coverage while maintaining tolerable dose levels to OARs. However, in certain cases, a clear compromise arises between achieving optimal target coverage and limiting exposure to adjacent critical structures. In these instances, other considerations with both pros and cons may be taken into account like mastectomy instead of BCS plus RT [23], omitting RT after BCS [24], partial breast irradiation when using [25], respiratory gating [26], prone position [27], intensity modulated RT 3DCRT [28], and proton RT [29]. Various strategies exist to reduce radiation exposure to the heart and coronary arteries; however, some of these techniques can be costly, time-intensive, and may not be accessible to the potentially large population of women undergoing breast cancer treatment. Therefore, studies employing a risk-benefit and cost-effectiveness approach are necessary to identify patients who would benefit from alternatives to 3DCRT. In our study, we aimed to evaluate the changes in ejection fraction before and after radiotherapy, in order to assess the cardiac impact of 3DCRT in patients treated at the Nuclear and Radiotherapy Center of Baghdad Medical City.
Patients and methods
The study is a prospective observation study, which was done in August 2016 till January 2017(i.e. four months after finishing RT).
The study was conducted in The Nuclear and RT Center of the Oncology Teaching Hospital in Baghdad Medical City.
We collected the data from the files of fifty female patients of BC whom underwent surgery then systemic treatment followed by 3D conformal RT.
The inclusion criteria was female patients with age was between (30- 79) years old, with good performance status (ECOG=0-1), positive lymph nodes. Patients who were HER2-negative or had a history of IHD, HTN, or DM were excluded from the study.
The study depends on the size of the Ejection fraction measured by ECHO study before and four months after RT.
The EF: is the percentage representing the ratio of the volume of blood the heart pumps out during systole to the total volume of blood in the heart at the end of diastole, typically ranging between 50-80%.
Statistical analysis
SPSS version 20 was used for data entry and analysis and results were expressed as mean and SD. Paired sample and independent student t test were used to confirm significance, p<0.05 was considered significant.
Results
A total of 50 patients with age ranged from 32-72 years (mean age=53±11.3 SD) were enrolled in this study. The highest percentage (28%) of cases were in age group of 40-49 years, 26% were in age group of 60-90 years, 24% were in age group of 50-59% and the remainders of cases were distributed in different percentages to others age groups. The finding of current study revealed that52% of cases were involved the right breast and 56% of cases were underwent conservative surgery (Table 1).
The findings of current study revealed there was statistical significant difference with regards to mean value of ejection fraction pre and post radiation with 4260cGY and 5000cGY (p=0.001). On further analysis of data according to site of involvement the significant difference was reported with 5000 Gy dose in both sites (Table 2).
The results demonstrated there was no significant difference (p=0.2) in mean value of ejection fraction (post radiation) of patients who were irradiated by 4260 or 5000 Gy (Table 3).
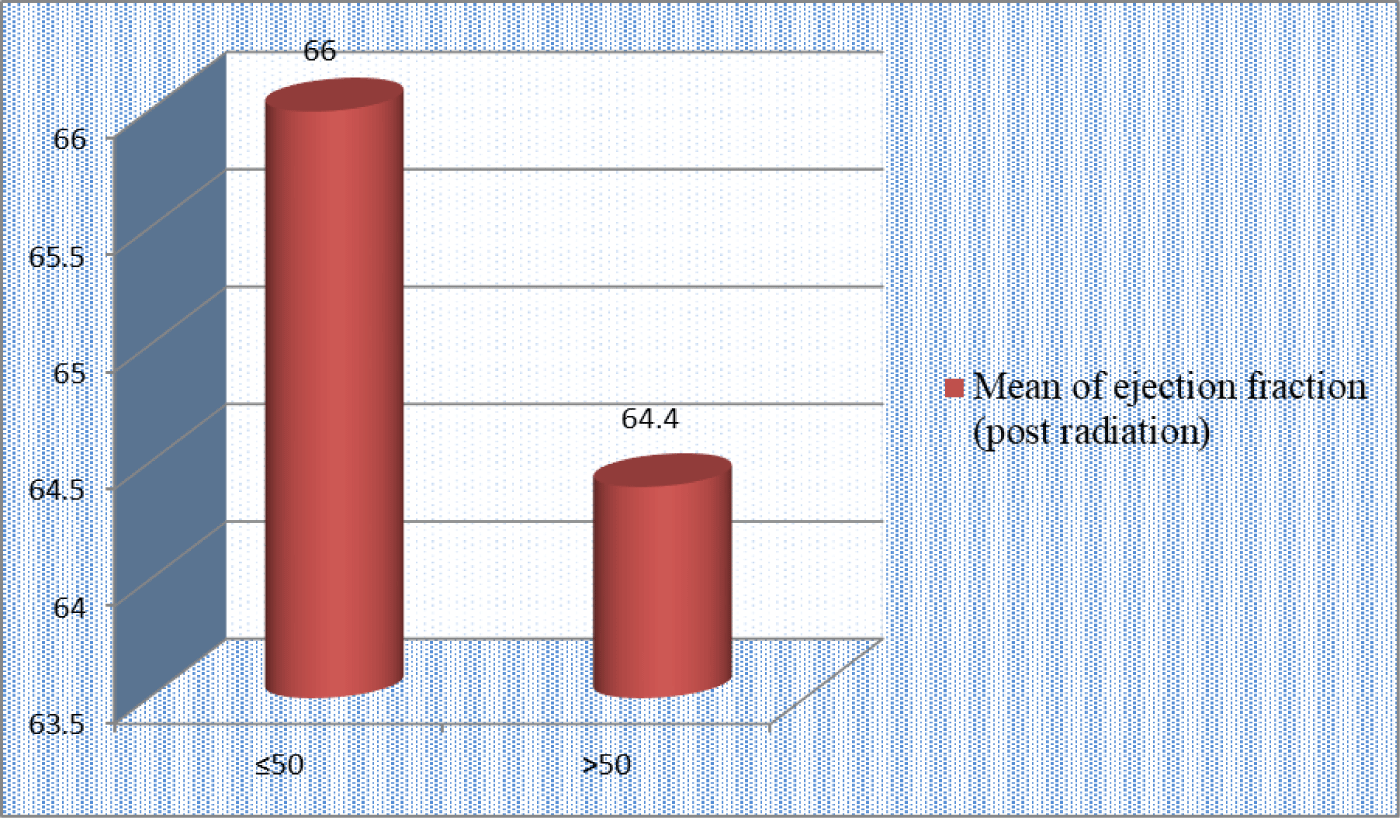
The results of analysis of mean value of post radiations ejection fraction according to age of the patients whatever the dose of radiation who were received showed there was no significant difference in mean value of ejection fraction between the age category of the patients (p≥0.1) (Figure 1).
Discussion
The earliest evidence of radiation-induced cardiac toxicity came from the Stockholm breast cancer trial, which delivered 45 Gy at 1.8 Gy per fraction and reported a 15-year excess cardiac mortality of 6.8% attributed to radiation exposure. A subsequent review of patients from this trial estimated that, on average, 25% of the heart volume received at least 22.5 Gy (V22.5 < 25%). One of the pioneering studies demonstrating a method to reduce heart dose involved analyzing treatment plans of 100 women with left-sided T1N0M0 breast cancer who underwent lumpectomy followed by three-dimensional conformal radiation therapy (3DCRT) at 50 Gy in 2 Gy fractions. This review found that the volume of the heart receiving 50% of the prescribed dose was reduced to approximately 5.7%, or around 33 cc. The substantial decrease in cardiac radiation dose contributed to the widespread adoption of 3D conformal radiation planning in breast cancer treatment [30].
Seddon, et al., compared radiation-induced myocardial perfusion defects using SPECT imaging in patients with early-stage left- versus right-sided breast cancer treated with modern radiotherapy techniques. They found that left-sided breast cancer was associated with a significantly higher number of myocardial perfusion abnormalities compared to right-sided cases. These defects included both reversible and irreversible changes, indicating that radiotherapy can cause myocardial injury as well as epicardial coronary disease. However, after a minimum follow-up of five years post-treatment, none of the detected abnormalities were deemed clinically significant [31].
Darby, et al., demonstrated that exposure of the heart to ionizing radiation during breast cancer radiotherapy increases the risk of ischemic heart disease. This risk rises proportionally with the mean heart dose, becoming evident within a few years after treatment and persisting for at least two decades. Women with existing cardiac risk factors experience a greater absolute increase in risk compared to those without such factors [32].
Conversely, Dr. Harris highlighted that despite this dose-response relationship, the absolute risk increase remains small. For instance, in 50-year-old women without preexisting cardiac conditions, the lifetime risk increases were estimated at 0.5% after a 0.5 G dose, 0.2% after 1 G, and 0.5% after 3 G to the heart. Currently, most node-negative women undergoing breast-conserving therapy receive a mean heart dose of approximately 1 G, while slightly higher doses (around 2 G) are more common in left-sided post-mastectomy radiotherapy. He emphasized that these contemporary heart doses confer a relatively minor mortality risk when weighed against the substantial survival benefits provided by RT [33].
In our study we had reviewed 50 patients with BC ,whom were in need for RT as part of their management, with 48% have left sided BC, 52% have right sided BC ,and 44% have chest wall irradiation (4260 cG/16F), while other 56% underwent whole breast irradiation (5000 cG/25F). The RT technique was in 3D conformal planning for both modalities.
Author’s contributions
1: Conceptualization; Data Curation; Methodology; Project administration; Resources; Writing – original draft and Writing – review & editing
2: Conceptualization; Data Curation; and Writing – review & editing
3: Conceptualization; Data Curation; supervisor; draft and Writing – review & editing
4: Conceptualization; Data Curation; soft were; Writing – original draft and Writing – review & editing
Funding
None.
Conflicts of interest
The authors declare no conflict of interest regarding this article.
References
- (2015) Manual of Clinical Oncology. (9th edn). Union for International Cancer Control. Wiley Blackwell 221.
- IRAQI CANCER REGISRTY (2011).
- Sarah C Darby, Marianne Ewertz, Paul McGale, et al. (2013) Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med 368: 987-998.
- Hirofumi Saiki, Ivy A Petersen, Christopher G Scott, et al. (2017) Risk of Heart Failure With Preserved Ejection Fraction in Older Women After Contemporary Radiotherapy for Breast Cancer. Circulation 135: 1388-1396.
- Bearzi C, Marcello Rota, Toru Hosoda, et al. (2007) Human cardiac stem cells. Proc Natl Acad Sci USA 104: 14068-14073.
- Fajardo LF, JR Stewart, KE Cohn (1968) Morphology of radiation induced heart disease. Arch Pathol 86: 512-519.
- Schultz-Hector S, KR Trott (2007) Radiation-induced cardiovascular diseases: is the epidemiologic evidence compatible with the radiobiologic data. Int J Radiat Oncol Biol Phys 67: 10-18.
- Neil K Taunk, Bruce G Haffty, John B Kostis, et al. (2015) Radiation-Induced Heart Disease: Pathologic abnormalities and putative mechanisms. Front Oncol 5: 39.
- Stewart JR, L F Fajardo, S M Gillette, et al. (1995) Radiation injury to the heart. Int J Radiat Oncol Biol-Phys 31: 1205-1211.
- Veinot JP, WD Edwards (1996) Pathology of radiation-induced heart disease: A surgical and autopsy study of 27 cases. Hum Pathol 27: 766-773.
- Mulrooney DA, Mark W Yeazel, Toana Kawashima, et al. (2009) Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: Retrospective analysis of the Childhood Cancer Survivor Study cohort. BMJ 339: b4606.
- Schultz-Hector S, KR Trott (2007) Radiation-induced cardiovascular diseases: Is the epidemiologic evidence compatible with the radiobiologic data. Int J Radiat Oncol Biol Phys 67: 10-18.
- Benditt EP, JM Benditt (1973) Evidence for a monoclonal origin of human atherosclerotic plaques. Proc Natl Acad Sci USA 70: 1753-1756.
- Murry CE, C T Gipaya, T Bartosek, et al. (1997) Monoclonality of smooth muscle cells in human atherosclerosis. Am J Pathol 151: 697-705.
- Senkus-Konefka E, J Jassem (2007) Cardiovascular effects of breast cancer radiotherapy. Cancer Treat Rev 33: 578-593.
- Cuzick J, H Stewart, L Rutqvist, et al. (1994) Cause-specific mortality in long-term survivors of breast cancer who participated in trials of radiotherapy. J Clin Oncol 12: 447-453.
- Darby S, Paul McGale, Richard Peto, et al. (2003) Mortality from cardiovascular disease more than 10 years after radiotherapy for breast cancer: nationwide cohort study of 90 000 Swedish women. BMJ 326: 256-257.
- Roychoudhuri R, David Robinson, Venkata Putcha, et al. (2007) Increased cardiovascular mortality more than fifteen years after radiotherapy for breast cancer: a population-based study. BMC Cancer 7: 9.
- Darby SC, Paul McGale, Carolyn W Taylor, et al. (2005) Long-term mortality from heart disease and lung cancer after radiotherapy for early breast cancer: prospective cohort study of about 300,000 women in US SEER cancer registries. Lancet Oncol 6: 557-565.
- Prosnitz RG, Jessica L Hubbs, Elizabeth S Evans, et al. (2007) Prospective assessment of radiotherapy-associated cardiac toxicity in breast cancer patients: analysis of data 3 to 6 years after treatment. Cancer 110: 1840-1850.
- Wang W, Rodrigo Wainstein, Xavier Freixa, et al. (2011) Quantitative coronary angiography findings of patients who received previous breast radiotherapy. Radiother Oncol 100: 184-188.
- Clarke M, R Collins, S Darby, et al. (2005) Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: An overview of the randomised trials. Lancet 366: 2087-2106.
- Darby S, P McGale, C Correa, et al. (2011) Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 378: 1707-1716.
- Pignol JP, BM Keller, A Ravi (2011) Doses to internal organs for various breast radiation techniques--implications on the risk of secondary cancers & cardiomyopathy. Radiat Oncol 6: 5.
- Valachis A, D Mauri, NP Polyzos, et al. (2010) Partial breast irradiation or whole breast radiotherapy for early breast cancer: a meta-analysis of randomized controlled trials. Breast J 16: 245-251.
- Vikstrom J, Mari H B Hjelstuen, Ingvil Mjaaland, et al. (2011) Cardiac and pulmonary dose reduction for tangentially irradiated breast cancer, utilizing deep inspiration breath-hold with audiovisual guidance, without compromising target coverage. Acta Oncol 50: 42-50.
- Chino JP, LB Marks (2008) Prone positioning causes the heart to be displaced anteriorly within the thorax: implications for breast cancer treatment. Int J Radiat Oncol Biol Phys 70: 916-920.
- Lohr F, et al. (2009) Potential effect of robust and simple IMRT approach for left-sided breast cancer on cardiac mortality. Int J Radiat Oncol Biol Phys 74: 73-80.
- Weber DC, Carmen Ares, Antony J Lomax, et al. (2006) Radiation therapy planning with photons and protons for early and advanced breast cancer: An overview. Radiat Oncol 1: 22.
- Robert E Beck, Leonard Kim, Ning J Yue, et al. (2014) Treatment Techniques to Reduce Cardiac Irradiation for Breast Cancer Patients Treated with Breast-Conserving Surgery and Radiation Therapy: A Review. Front Oncol 4: 327.
- http://www.sciencedirect.com/science/article/pii/S0167814012000680#b0165
- https://www.ncbi.nlm.nih.gov/pubmed/?term=N+Engl+J+Med+368%3A987-998%2C+2013
- http://www.ascopost.com/issues/october-15-2013/breast-cancer-radiotherapy-and-carodiotoxicity-what-is-the-true-risk/
Corresponding Author
Ahmed Alshewered, Misan Radiation Oncology Center, Misan Health Directorate, Ministry of Health, Misan, Iraq
Copyright
© 2025 Dakhil Ali Alasadi ZM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.