Mercury Levels in Human Hairs and Nails of Traditional Gold Miners in Butana-Area East of Sudan
Abstract
The aim of this study is to assess the mercury (Hg) exposure level of gold miners in Butana mining area, in Eastern, Sudan. To achieve this goal 40 biological samples such as hair and nails (20 for each) samples were obtained randomly from the gold miners in 10 sampling sites and analyzing using US EPA Method 7473. A direct mercury analyzer (DMA-80; Milestone Inc., Shelton, CT, USA) was adopted to determine the mercury levels. The average of mercury concentration levels in the hair samples of gold miners were 8.34 mg/kg, 5.14 mg/kg, 7.26 mg/kg, 4.40 mg/kg, 3.04 mg/kg, 4.81 mg/kg, 3.49 mg/kg, 4.29 mg/kg, 3.26 mg/kg, 3.02 mg/kg respectively, while in nail samples were 7.18 mg/kg, 6.78 mg/kg, 7.02 mg/kg, 4.40 mg/kg, 4.39 mg/kg, 7.26 mg/kg, 6.95 mg/kg, 2.53 mg/kg, 3.82 mg/kg, 3.55 mg/kg respectively. The results indicated that the mercury levels in all samples exceeded the permissible limit set by Environmental protection agency, (USEPA) and World health organization, (WHO). The research indicates that the gold miners and local populations in the study area can have high exposure to mercury vapors because the miners burn the gold-mercury amalgam to vaporized the mercury and recover the gold. This mercury exposure has caused disease such as heart, respiratory, kidney, joints and skin problem, Cough, sore throat Shortness of breath, Nausea, vomiting, diarrhea, Increase in blood pressure or heart rate.
Keywords
Mercury, Butana mining area, Biological samples, Hair, Nails, US EPA Method 7473, Direct mercury analyzer
Introduction
Mercury (Hg) is a naturally occurring element found in air, water, and soil. It is distributed throughout the environment by both natural and anthropogenic (human) processes. Mercury is found in various inorganic and organic forms and is persistent in the environment. The three predominant forms include: a) Elemental mercury (with the chemical symbol of Hg0; b) Ionic mercury (also known as inorganic mercury with the chemical symbol of Hg (II) or Hg2+) which in nature exists as Hg (II) mercuric compounds or complexes in solution; and c) Organic mercury with methyl mercury (with the chemical symbol of MeHg) being the most important, while most of the mercury in water, sediments, soil, plants and animals as found inorganic and organic forms of the element.
Mercury is considered as one of the toxic and dangerous heavy metals [1,2], Hg can easily accumulate in the food chain and reaches to the human body through ingestion pathway and can also easily enter in to human body through inhalation and others routes and causes harmful effects on human health [3]. People may be exposed to elemental or inorganic mercury through inhalation of ambient air during occupational activities, occupational exposures can occur where mercury or mercury compounds are produced, used in processes, or incorporated in products. Occupational exposures it is reported from mercury mines, mercury-based small-scale gold and silver mining, refineries, chlor-alkali plants and others.
For centuries, the capacity exhibited by mercury to form amalgamates with gold has led people in Small-scale or artisanal mining, using gold-mercury amalgamation to extract gold from ore. Miners burn the gold-mercury amalgam to vaporized the mercury and recover the gold, thus the miners and local populations can have high exposure to mercury vapors. Mercury releases occur as liquid mercury lost to aquatic environments during the amalgamation process or in waste discharges and vapors entering the atmosphere. Exposures can be estimated by measuring pollutant levels like mercury in various body tissues (such as hair, blood, cord, urine, human milk and nails). These measurements, also known as biological markers (or biomarkers), are useful tools for human exposure assessment [4]. Analyzing mercury in biological samples is complicated by the different organic and inorganic forms of the metal that may be present. Therefore, all the mercury in the sample is usually reduced to its elemental state prior to analysis. Mercury is also relatively volatile and, therefore, easily lost during sample preparation and analysis. In spite of these complications, several methods have been developed for determining trace amounts of mercury in biological samples [5]. Most of the methods available to determine mercury levels in blood, urine, tissues, and hair use atomic absorption spectrometry (AAS), atomic emission spectrometry (AES), atomic fluorescence spectrometry (AFS), neutron activation analysis (NAA), mass spectrometry (MS), or anodic stripping voltammetry (ASV). Cold vapor AAS (or CVAAS) is the most widely used. In the past several researchers have focused on Hg contamination and bioaccumulation and its hazardous effects on human health [6,7]. Khan have previously worked on Hg concentrations in the blood and urine of workers involved in small-scale gold panning and extraction in the northern Pakistan but not focused on Hg contamination in their hair and nails [8]. To best of our knowledge this, the first study aiming to investigate the total Hg in the hair and nails samples of gold miners, in Butana mining area, in Eastern, Sudan.
Materials and Methods
Study site
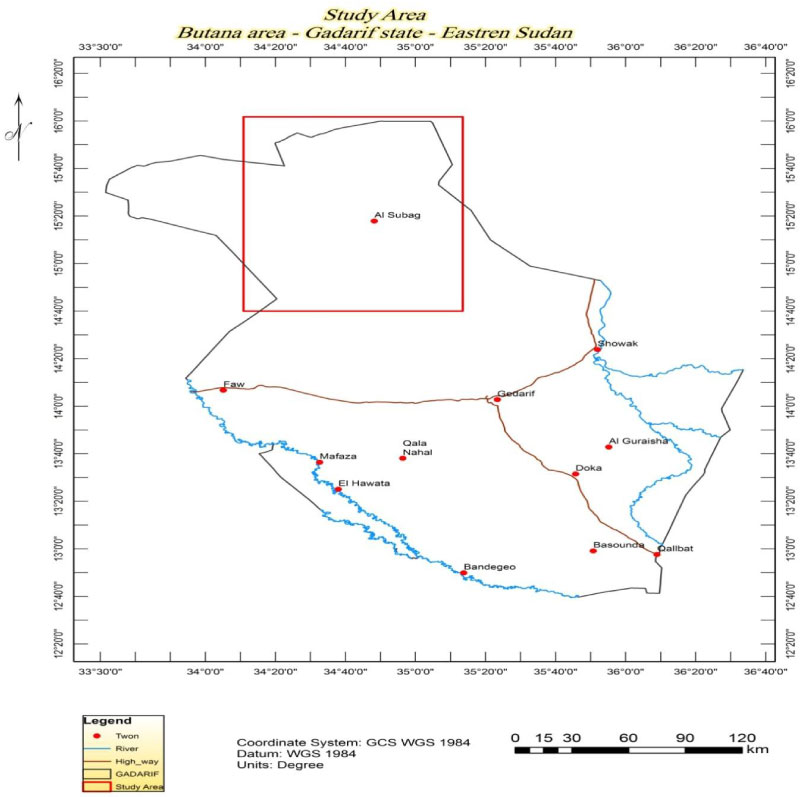
The study area is located in central Butana area Figure 1 within the administrative boundaries of rural Subagh locality of Gedarif State, Eastern Sudan, it comprises vast clay plains extending from the Gedarif area in the south up to latitude -15°N and from the banks of the Blue Nile River in the west up to banks of the Atbara River in the east. Climatically, the area is set within semi-arid to Savannah region [9] with high temperature in summer is around 40-450C (March - October) and cold winter 18-25 ℃ in winter (December-February).
The vegetation is spares, confined to the valleys and is made up of acacia type (Acacia Sayal and Acacia mallifera). The rainy season in the Central Butana is between 150 to 400 mm during the months of July and September, raining although intermittent increases from north to south. There are two main facilities for surface water storage in the study area. The Hafirs are very common water reservoirs throughout the Sudan. They are water harvest catch-up of rain water in the form of small dams. They are the only sources of water supply for human and animal consumption. The other source of water supply is ground water, in which wells are scattered through far distances.
Gold mining
The study area contains gold and minerals mines. Heaps of metal ores including precious, strategic, base metals and industrial minerals were scattered all over the area. The most important: Precious metals such as gold, silver and platinum, Base metals such as copper zinc and lead, strategic minerals such as iron, manganese, chromium and pentanium, Industrial metals such as calcium carbonate, salt, talc and kaolin, silicon, and zircon and Building stones. Butana area contains many locally named mines like: Um Sarha, Fuwail, Khiari, Wad Bushara, Shawor, Gilaitah, Ead AL Wahash, and Birasi.
Sampling procedure
A total of 20 hair samples and 20 nails samples were collected from 10 sites in Butana miming area, Random sampling was adopted. Sampling has done between May and June 2018. The samples of hair were taken from occipital region of the head, using clean stainless steel scissors. Once cut, the hair bundle was wrapped closed to the scalp end with a clean piece of paper and held together with a plastic clip. The Samples of nails were cut using clean stainless steel scissors, then was wrapped in a clean piece of paper and held together with a plastic clip. All samples were packed in clean, sealable bags and transferred to the laboratory for analysis.
Preparation of standards
The standard solutions of mercury were prepared by serial dilution from known Standard stock solutions of 1000 mg/L. A calibration curve was prepared and then the analysis of the samples for the mercury was performed.
Determination of the concentration levels of mercury
The total-Hg in the hair and nails samples were determined using US EPA Method 7473 and a direct mercury analyzer (DMA-80; Milestone Inc., Shelton, CT, USA). The method is based on thermal decomposition, amalgamation, and AAS. Sample (0.009 g) of hair samples and (0.001 g) of nails samples were weighed onto nickel boat, the boat was e placed into an autos ampler which sequentially inserts them into the combustion tube of the instrument. There, the samples were heated with oxygen flowing over them at 200 mL min-1, the decomposition and combustion products were swept through a catalyst tube where oxidation was completed, and nitrogen and sulfur oxides and halogens were trapped. The remaining gases, including Hg0, were carried to a gold amalgamator which selectively traps Hg. Later, the gold trap was rapidly heated releasing Hg vapor into the spectrophotometer. Absorbance, measured at 253.7 nm, is a function of Hg concentration. The parameters of DMA-80 direct mercury analyzer were adjusted according to the manufacturer's recommendation. The results were recorded in Table 1 and Table 2.
DMA-80 direct mercury analyzer
Milestone's DMA-80 is a direct mercury analyzer, which uses the principle of thermal decomposition, amalgamation and atomic absorption. Analysis takes only 5 minutes per sample and does not require any sample preparation, The DMA-80 is used for analysis environmental, geochemical, petrochemical, food and Feed, clinical and polymer samples.
Principle of operation
The sample introduction device consists of a motorized support equipped with a metal or metal alloy sample boat. Once the sample is either manually or automatically dispensed into the sample boat, the boat is mechanically introduced automatically into a quartz decomposition tube. The decomposition tube is heated by two independently programmable furnaces -the decomposition and catalyst furnaces - and each furnace is capable of maintaining a temperature of at least 750 EC. The samples are first dried and then thermally decomposed in an oxygen rich furnace, releasing mercury vapor.
Mercury and other combustion products are released from the sample and they are carried to the catalyst section of the furnace, where nitrogen and sulfur oxides, as well as halogens and other interfering compounds, are eliminated.
The mercury vapor is transported by oxygen over the amalgamator that traps the mercury. Mercury is selectively trapped, in a separate furnace, through gold amalgamation. The amalgamation furnace is heated and mercury is rapidly released. The mercury vapor flown via the carrier gas into a unique block with tri-cell arrangement, positioned along the optical path of the spectrophotometer, where it is quantitatively measured by atomic absorption at 253,65 nm. The flow path through the spectrometer and cuvettes is maintained at approximately 120 EC, by a heating unit, to prevent condensation and minimize carry-over effects. A mercury vapor lamp is used as the light source. The detector is connected to a computer for data acquisition and analysis.
Questionnaire survey
To the volunteers of the research (miners) we applied questionnaires about the living standards, gender, age, education, habits, interests, activities, health, weight, jobs, eating pattern, common diseases, uses of drug or not, drinking water sources, other water sources and its uses, about gold mining and their experience.
Results and Discussion
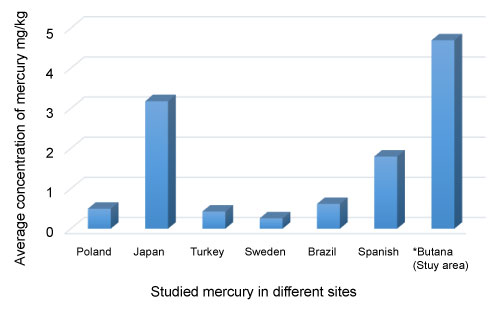
This is the first research work conducted in the study area to investigate the contamination of Hg in the biological samples (hair and nails) collected from gold miners and its adverse impacts on the miners' health. Gold mining has attracted the attention worldwide now a day because of the uses of Hg and its adverse impacts on environment including all biological resources. Many researchers showed the contamination of Hg in biological samples of humans of different areas of world due to gold mining [10,11]. Hair is the preferred choice for many studies as it provides a simple, integrative, and non-invasive sample for estimating long-term average exposure, although is not as good an indicator of mercury vapor exposure as urine is [4,12]. Total mercury in hair is about 250 to 300 times higher than the blood mercury concentration at the moment hair is formed., According to WHO [13], The normal level of mercury in hair is 1-2 ppm (or 1-2 mg/kg), the people without Hg exposure have Hg level ≤ 5 μg/kg in their blood. Small exposure contaminated with Hg has led to 10-20 µg/kg of Hg, while heavy exposure has caused Hg contamination in the range from 100 to 200 µg/kg in the users' blood, hair had also contaminated with Hg at range of 20-50 µg/kg [14]. Measuring the concentrations of mercury in hair and nails as biomarker for short term and long- term exposures gave an insight into the levels of exposure for the miners. The results in Table 1 and Table 2 show that the miners who are in the study area are exposed to great danger of contamination get to severe poisonings in their health resulted from heating metallic mercury and release mercury vapor at levels approaching occupational threshold limit values. US- Environmental Protection Agency (US- EPA) [15] recommended guidelines and other countries (Poland, Japan, Turkey, Sweden, Brazil and Spanish) for maximum levels for Hg in hair and nail samples.
The Average concentrations of mercury in hair sample ranged from 3.02 to 8.34 mg/kg. The results indicated that Hg in hair samples at different sampling sites exceeded the permissible limit 1 mg/kg as the Threshold level according to US- Environmental Protection Agency (US- EPA) and World Health Organization (WHO). Mercury recorded the highest concentrations in W.Bushara site and the lowest concentration in Wadi-Al Rawyan site. For comparison, the concentration level of mercury at the different sites in the study area was compared with the mercury level in different countries, Table 3 [16-21].
According to Table 2, the concentration level of mercury in hair samples of study area (4.70 mg/kg) presented the highest concentration comparing with the levels of mercury in different countries, while mercury concentration in Sweden presented the lowest: mercury concentration in study area was almost 18 times as much as that of mercury in Sweden country Figure 2. This show long term exposure occurred through using gold-mercury amalgamation to extract gold from ore and dangerous effect of work in roasting of amalgamated gold and inhalation the air. Therefore, the exposure levels of mercury to the miners in study area should be paid more attention to in the future.
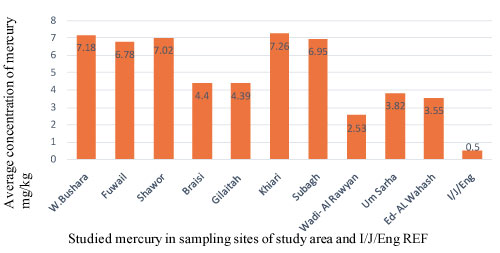
The average concentrations of mercury in nail samples of gold miners ranged from 2.53 to 7.26 mg/kg. Mercury recorded the highest mean concentrations in Khiari site and the lowest concentration in in Wadi-Al Rawyan site.
The analyzed samples of nails in Figure 3 showed the mercury concentration at different sampling sites is highest than the maximum permissible levels of Italy/Japan/England.
Conclusions and Recommendation
In conclusion, the concentrations of the mercury in hair samples in all sampling sites exceeded the mercury threshold level according to US- Environmental Protection Agency (US- EPA) and World Health Organization (WHO), and highest than the mercury concentration level in different countries and level of mercury in nail samples in all sampling sites above the maximum permissible levels of Italy/Japan/England.
The recorded high levels of mercury concentrations indicate that over time the gold miners and local population may accumulate extremely high levels of mercury through burn the gold-mercury amalgam to vaporized the mercury and recover the gold. Further, dependence of gold mining in this area has caused both human and environmental health problems, because that in order to safeguard the livelihood of people around mining sites, the Government should play their role, start awareness programs, Continued education on safety measures concerning the human and environmental health, Active education on geological related illnesses or disorders, Encouragement to use of protective gear at all times during gold mining and retorts, simple fume chambers, etc. during processing; Furthermore, proper legislation relating to gold mining to be put in place to check against illegal mining activities that degrade the environment through inappropriate mining to protect them from the use and effects of mercury.
Acknowledgement
Our thanks are due to the Ministry of minerals - Geological Research Authority (GRAS) for funding the research project. Thanks are also due to The State of Gedarif local Government Office for facilitating the tours around the sites in which the work has been carried out. Our thanks and gratitude is due the University of Medical Sciences and Technology and University of Khartoum for the supervision of this research project.
References
- Jia X, Gonga D, Hana YB, et al. (2012) Fast speciation of mercury in seawater by short-column high-performance liquid chromatography hyphenated to inductively coupled plasma spectrometry after online cation exchange column preconcentration. Talanta 88: 724-729.
- Dryzalowska A, Falandysz J (2014) Bioconcentration of mercury by mushroom Xerocomuschrysenteron from the spatially distinct locations: Levels, possible intake and safety. Ecotoxicol Environ Saf 107: 97-102.
- Zheng L, Liu G, Chou C (2007) The distribution, occurrence and environmental effect of mercury in Chinese coals. Sci Total Environ 384: 374-383.
- International Programme on Chemical Safety (IPCS) (2000) Environmental health criteria 214, human exposure assessment. World Health Organization, Geneva.
- Agency for Toxic Substances and Disease Registry (ATSDR) (1999) Toxicological profile for mercury. US Department of Health and Human Services, Public Health Service, ATSDR, Atlanta, Georgia.
- Eisler R (2002) Health risks of gold miners: A synoptic review, US geological survey. Environ Geochem Health 25: 325-345.
- Veiga MM, Maxson PA, Hylander LD (2006) Origin and consumption of mercury in small-scale gold mining. J Cleaner Prod 14: 436-447.
- Khan S, Shah MT, Din I, et al. (2012) Mercury exposure of workers and health problems related with small-scale gold panning and extraction. J Chem Soc Pak 34: 870-876.
- Van Der Kevie W (1973) Climate Zones in the Sudan. Soil Survey Department Wad medani.
- Straaten PV (2000) Human exposure to mercury due to small scale gold mining in northern Tanzania. Sci Total Environ 259: 45-53.
- Cortes-Maramba N, Reyes JP, Francisco-Rivera AT, et al. (2006) Health and environmental assessment of mercuryexposure in a gold mining community in Western Mindanao, Philippines. J Environ Manag 81: 126-134.
- United Nations Industrial Development Organization (UNIDO) (2003) Removal of barriers to introduction of cleaner artisanal gold mining and extraction technologies.
- WHO (2003) Elemental mercury and inorganic mercury compound: Human health aspects. In: CICAD, World Health Organization, WHO, Geneva.
- Adimado AA, Baah DA (2002) Mercury in human blood, urine, hair, nails and fish from the Ankobra and Tano river basins in Southwestern Ghana. Bull Environ Contam Toxicol 68: 339-346.
- USEPA (1997) Mercury study report to congress. United State Environmental Protection Agency, EPA-452/R-97-003.
- Chojnacka K, Gorecka H, Chojnacki A, et al. (2005) Inter-element interactions in human hair. Environ Toxicol Pharmacol 20: 368-374.
- Sera K, Futatsugawa S, Murao S (2002) Quantitative analysis of untreated hair samples for monitoring human exposure to heavy metals. Nucl Instrum Methods B 189: 174-179.
- Do gan-Sa glamtimur N, Kumbur H (2010) Metals (Hg, Pb, Cu, and Zn) bioaccumulation in sediment, fish, and human scalp hair: A case study from the city of Mersin along the southern coast of Turkey. Biol Trace Elem Res 136: 55-70.
- Rodushkin I, Axelsson MD (2000) Application of double focusing sector field ICP-MS for multielemental characterization of human hair and nails. Part II. A study of the inhabitants of northern Sweden. Sci Total Environ 262: 21-36.
- Miekeley N, Dias Carneiro M, da Silveira CLP (1998) How reliable are human hair reference intervals for traceelements? Sci Total Environ 218: 9-17.
- Gonzalez-Munoz MJ, Pen A, Meseguer I (2008) Monitoring heavy metal contents in food and hair in a sample of young Spanish subjects. Food Chem. Toxicol 46: 3048-3052.
Corresponding Author
Abdalla A Elbashir, Chemistry Department, Faculty of Science, University of Khartoum, Sudan.
Copyright
© 2019 Idris IM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.